The term “butterfly children” sounds pleasant but refers to a harsh reality: children with epidermolysis bullosa, or EB, an inherited, genetic blistering skin disorder, are born with skin as fragile as a butterfly’s wings.
EB affects all races, ethnicities and both genders, and has no cure. According to the Dystrophic Epidermolysis Bullosa Research Association of America, every year about 200 children are born with EB in the United States.
There are several types of EB. The most common type of severe EB is recessive dystrophic epidermolysis bullosa, or RDEB—a progressive, devastatingly painful and debilitating disease that affects up to ~2,500 patients in the U.S. and often leads to death. RDEB causes severe blistering and areas of missing skin in response to any kind of friction, including normal rubbing and scratching.
Cause of RDEB
RDEB is an autosomal recessive, inherited skin disease caused by null mutations within the type VII collagen gene (COL7A1). The mutations cause an absence or reduction of functional collagen VII (COL7), which make up anchoring fibrils that maintain binding of the epidermis to the dermis. The disease is characterized by a mechanical fragility and repeated blister formation in the sub-lamina densa at the level of the structurally defective anchoring fibrils.
Development of a New Gene Therapy
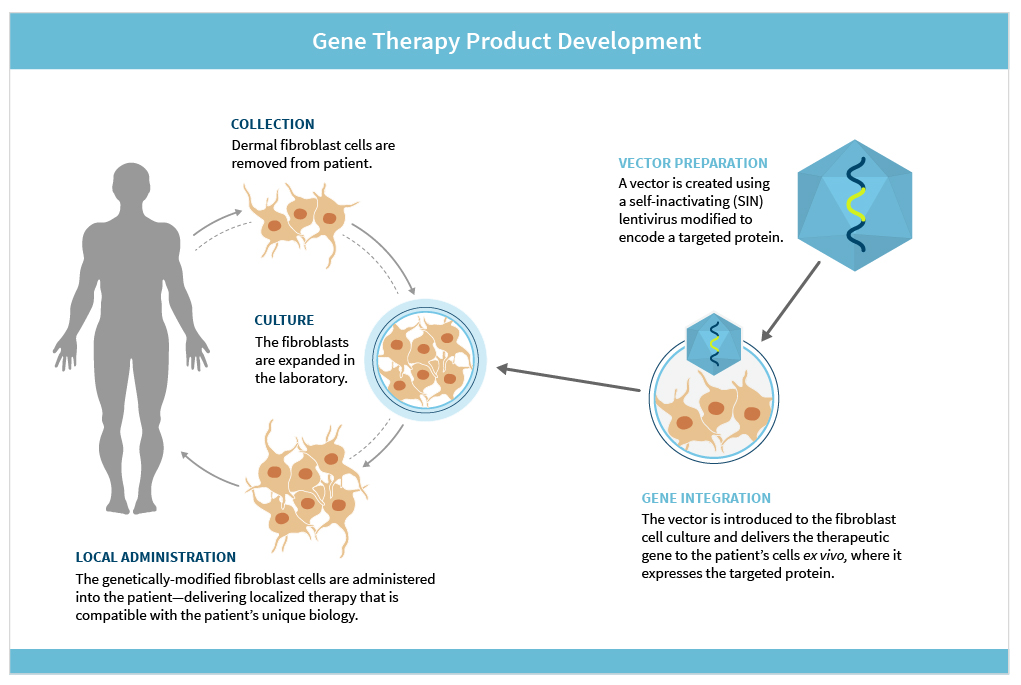
Fibrocell, a gene therapy company, is applying its distinctive autologous fibroblast technology to develop FCX-007, its candidate for the treatment of RDEB—and potentially the first investigational therapy to target the underlying cause of the disease.
FCX-007 is being developed in collaboration with Intrexon Corporation, a leader in synthetic biology.
FCX-007 is an autologous dermal fibroblast genetically modified to express functional COL7 that is missing or deficient in RDEB patients. Transduced with a lentiviral vector containing COL7A1, FCX-007 is injected directly into the papillary dermis of blisters and wounds where the COL7 protein could enable formation of anchoring fibrils to hold the layers of skin together. The goal of the therapy is to provide high levels of functional COL7 directly to the affected areas while avoiding systemic distribution. This localized therapeutic approach is compatible with the unique biology of each individual patient.
The U.S. Food and Drug Administration (FDA) has granted Orphan Drug Designation to FCX-007 for the treatment of Dystrophic Epidermolysis Bullosa, which includes RDEB; likewise, the FDA granted both Rare Pediatric Disease and Fast Track Designations to FCX-007 for the treatment of RDEB.
A poster presented at the American Society of Human Genetics Annual Meeting in October 2015 addressed the preclinical development of FCX-007 for the treatment of RDEB. As noted in the poster, in vitro product development data indicates that cGMP scale FCX-007 cells express full-length COL7 exhibiting the proper trimeric structure, size and binding functionality.
Production of the lentiviral vector (LV-COL7) was successful, resulting in an infectious titer of ~9 x 106 IU/mL. The integrated transgene copy number per cell was dependent on the virus dose. FCX-007 was produced by expanding fibroblasts from skin biopsies, transducing with the vector, followed by subsequent expansion, harvest and cryopreservation. The COL7 expression from the FCX-007 cells was confirmed by ELISA and Western Blot as well as qRT-PCR and immunofluorescence staining.
The structure of expressed COL7 was confirmed to be predominantly trimeric by immunoprecipitation/SDS-PAGE/Western blot analysis. The COL7 produced from the FCX-007 cells was demonstrated to be functional by binding to Laminin332 in an in vitro binding assay as well as by correction of the hypermotility phenotype of RDEB cells in an in vitro migration assay.
The presentation also reported in vivo results from a preclinical animal model evaluating FCX-007 in RDEB and normal human skin xenografts implanted onto the dorsum of immunodeficient SCID mice. The grafts consisted of human fibroblasts and keratinocytes in a bilayer. The goals of the study were to confirm persistence, distribution and localization of COL7, and to evaluate any potential for product toxicity or vector biodistribution.
The composite RDEB skin grafts were injected intradermally with 1 x 106 FCX-007 cells and analyzed by immunofluorescent staining with human COL7 specific antibodies. Localization of COL7 was observed in composite grafts 10 days post-injection. Positive control grafts generated from normal keratinocytes and fibroblasts showed intense COL7 staining and negative control grafts did not show COL7 staining at baseline measurements.
In a follow-up toxicology/biodistribution study, FCX-007 was well tolerated up to six months post-administration.
Phase 1/2 Clinical Trial Underway
Fibrocell has initiated a Phase 1/2 clinical trial of FCX-007 for the treatment of RDEB. The primary objective of this open-label clinical trial is to evaluate the safety of FCX-007 in RDEB patients. Additionally, the trial will assess the mechanism of action of FCX-007 through the evaluation of COL7 and the presence of anchoring fibrils, and the efficacy of FCX-007 through evidence of wound healing.
The first adult patient was dosed in the Phase 1 portion of the Phase 1/2 trial in February 2017. Prior to treatment of additional adult patients, the clinical trial protocol requires that a Data Safety Monitoring Board (DSMB) assess safety data from the first patient four weeks after administration of FCX-007. The data showed there were no product-related adverse events. As a result of this planned review, the DSMB allowed the continuation of dosing in May 2017.
Prior to conducting clinical trials on pediatric patients, Fibrocell is required to obtain allowance from the FDA by submitting evidence of FCX-007 safety and benefit in adult patients and data from its completed preclinical toxicology study.
With no FDA-approved treatments for RDEB, Fibrocell believes its gene therapy candidate, FCX-007, could become a significant advance in RDEB patient care.
John Maslowski is Chief Executive Officer of Fibrocell, an Exton, PA-based gene therapy company focused on transformational autologous cell-based therapies for rare skin and connective tissue diseases.