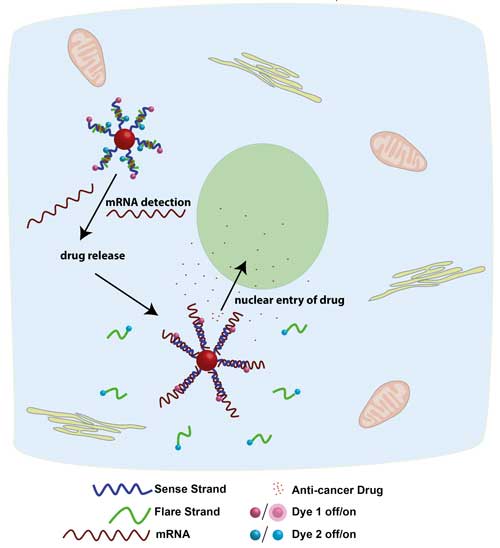
Schematic illustrating the design of the smart nanoparticle and its function. The sense and the flare strand form the short drug-loaded duplex on the surface of the gold nanoparticle. In the presence of the appropriate mRNA, both the flare strand and the drug are released inside the cell. The drug enters the nucleus of the cell and there results in cell death. Image: Dr. Amelie Heuer-Jungemann
Scientists from the University of Southampton designed an advanced type of nanoparticle, which is able to carry drugs directly into cells and release them only in the presence of an appropriate mRNA signature; in other words, the nanoparticle carriers release their payload only in specific — metastatic cancer — cells and remain inactive in healthy cells.
In their recent publication in Nanoscale (“Selective killing of cells triggered by their mRNA signature in the presence of smart nanoparticles”), the scientists designed nanoparticles that can selectively distinguish healthy cells from model metastatic cells and release their payload — an anticancer drug — only to the model metastatic cells.
The nanoparticle core is made of gold and it is coated with short duplex DNA strands, in which the anticancer drug doxorubicin is intercalated. Once the appropriate messenger RNA (a critical biomolecule for the production of proteins) is detected in the model cancer cells the DNA duplex dehybridizes and the drug is released.
This can be monitored in real time through an onset of fluorescence from the dehybridized DNA strand.
The most important aspect of this technology is that the release of the drug happens only in the presence of the appropriate mRNA. Thus, the drug is not released in the healthy cells, which do not contain the target mRNA.
“As far as we know this is the first example of nanoparticles that can selectively choose which cells to deliver their cargo to,” says Dr. Antonios Kanaras, head of the Laboratory for Inorganic Colloidal Nanocrystals and Applications group, which developed this new technology. “This is an important step forward for the development of new types of smart nanotechnological drugs. The use of smart nanoparticle designs, which release drugs selectively, can ensure minimal damage to healthy cells, be more efficient in smaller doses, and eliminate toxic side effects during the duration of a therapy (e.g. chemotherapy).”
The researchers chose a model cancer system to show that the drug can be selectively delivered to model metastatic cells. While all cells contained the nanoparticles, drug release was specific to the model metastatic cells.
“The beauty of these nanoparticles is that they essentially act as Trojan horses,” notes Dr. Amelie Heuer-Jungemann, the researcher who carried out the experiments. “They can be transported through several different cells without an effect, but if the correct trigger is present (i.e. the appropriate mRNA), the system steps into action, releases the drug and kills the cell. However, we could also imagine these nanoparticles to act as positive agents, for example releasing a drug that helps to keep the cell healthy rather than kill it. The possibilities of what we can do with them are really wide open!”
The team showed that by simply varying the sequence of the short DNA duplex attached to the gold nanoparticle, several different mRNAs can be detected and used to trigger the drug release in different cell types depending on their endocellular mRNA signature.
Future studies by the group will involve the incorporation of different drugs into the system as well as carrying out first studies in vivo.
These smart nanoparticles may have the potential to revolutionize (nano-)medicine and improve patient welfare.
Source: University of Southampton




