An extensive experiment testing the immune effects of a broad group of lab-designed nucleic acid nanoparticles did not find a strong, uniform immune response, as had been predicted. Instead, the tests found varying and specific responses from different immune cells, depending on each particle’s shape and formulation, a finding that may encourage further study of the particles’ therapeutic use.
In addition, the researchers believe that they may have discovered an “auxiliary” system for managing immune response — a molecular “alphabet” for communicating with the human immune system.
The findings were recently reported by Enping Hong, Ankit Shah, and Marina Dobrovolskaia of the Frederick Laboratory for Cancer Research, Emil Khisamutdinov of Ball State University and Justin Halman and Kirill Afonin of the University of North Carolina at Charlotte in the journal Nano Letters.
The idea has been around for a while that selected segments of RNA or DNA could be used therapeutically — therapeutic nucleic acids — to affect gene or cell function. Unfortunately, in clinical trials most of these proposed therapeutic molecules have proved to have an extreme side effect — they provoked a strong, often fatal reaction from the human body’s immune cells.
More recently, nanotechnologists have proposed designing self-assembling nanoparticles with the potentially therapeutic RNA or DNA sequences, combining the effects of several sequences into a targeted drug, giving multiple effects in a single particle, shaped into various designer forms — squares, triangles, cubes, and other structures. These potentially powerful therapeutic particles have been slow to be tested however, because researchers have theorized that they were likely to have the same “immunotoxic” effects as the natural nucleic acid fragments.
However, some nanoscientists questioned whether the projected immune reactions would necessarily be the case because of the complexities of immune system recognition and unique properties created by assembling traditional materials into nanoparticles of various shapes and structures.
“Even if nucleic acid nanoparticles are composed of components with known immunological toxicities, once you combine them and reformulate them, the nanoformulation becomes a completely different beast,” says Afonin, one of the paper’s corresponding authors.
“Our findings showed that, while some of the predictions were correct, many were completely wrong,” notes Afonin. “You can’t predict the immunotoxicity of nucleic acid nanoparticles purely by analyzing the responses to naturally produced DNAs and RNAs. We came up with some unexpected results.”
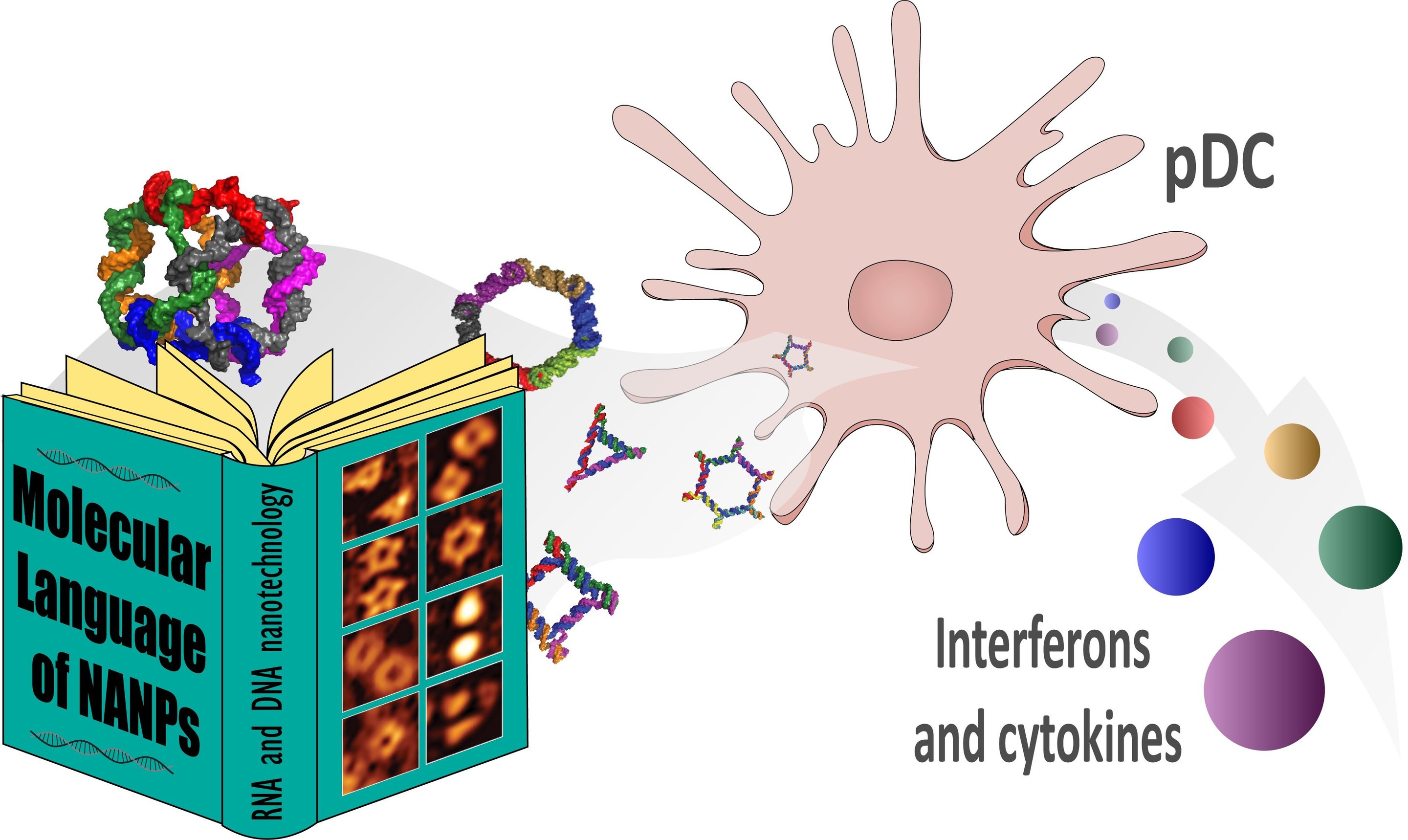
Immune response to nucleic acid nanoparticles provide a potential ‘alphabet’ for communicating with immune system cells. Credit: Reproduced with permission from Nano Lett. 2018, 18(7), 4309-4321. Image: Copyright 2018 American Chemical Society
To test the immunotoxicity of the particles and perhaps find clues for the mechanisms involved in immune cell response, Afonin and his colleagues selected a “library” of 25 different DNA or RNA nucleic acid nanoparticles designed by researchers in the field, carefully picked to “address all possible links” between their molecular properties and immune reactions. The library included a representative sampling of planar (flat), globular and fibrous (string-like) particles, with different sizes and molecular weights, as well as differing in a variety of critical chemical attributes. The particles were introduced to immune cells (peripheral blood mononuclear cells) from the blood of 60 unique human donors and monitored for the production of 29 different cytokines.
The details of the findings were revealing regarding the immunotoxicity of the particles because the immune response varied. But the results also revealed information regarding specific behavior of various immune cells.
A fundamental finding was that “naked” nucleic acid nanoparticles (unattached to other biological molecules) caused no immune response at all — because, the team found, unlike naturally occurring fragments of DNA or RNA, the constructed particles could not enter any immune cell without some kind of “carrier” molecules that allowed their entry. Effectively, plain nucleic acid nanoparticles are “invisible” to the human immune system.
Once the particles were paired with a carrier molecules, however, they were able to enter the cells, and elicited clear responses, as the researchers hoped. “The question is when we send this particle inside the human cell, what does the cell, particularly the immune cell, do?” Afonin says. “Does it see a particular shape as a threat?”
The results show that particle size, shape, 3-D structure (cubes, for example, compared with planar squares), DNA or RNA composition, and the chemical nature of how the particles were assembled (“connectivity”) all had distinctive effects on immune response and on which immune cells responded.
Among the details uncovered was the finding that particles composed of DNA tended to cause less immune response than those of RNA. RNA rings (flat structures) and RNA fibers caused less immune response than RNA cubes (globular structures). In further detail, DNA cubes induced the cytokine production of type I interferons alpha and omega, but only RNA cubes could induce type I interferon-beta or type III interferon-lambda. The different cytokines produced indicated that the differences in particles had a selective effect on that type of immune cell affected.
While the findings are scientifically important, the researchers stress that the new information has implications for future practical applications.
“Our findings highlight the key parameters that inform the way nucleic acid nanoparticles interact with the immune system,” the paper states. “These new insights improve the current understanding nucleic acid nanoparticles immunostimulatory properties, and pave the way to development of a new auxiliary molecular language that can be expressed through the script of rationally designed nucleic acid nanoparticles.”
“We have an alphabet to directly communicate with the immune system,” says Afonin. “Now we have to figure out the syntax of this new language; how to assemble these letters into the words, put sentences together, combine them into the paragraphs, and eventually, how to write a story. But right now we have an alphabet — it’s just the beginning, but I think this is fundamental work.”
Afonin points out that an “alphabet” that describes immune response to specific particle designs may naturally be useful for avoiding adverse reactions, but has more potential for situations where a response is actually desired (in the case of vaccines, for example) and has still more possibilities when treatment requires specific messaging to trigger a very specific immune response.
“If you need to deliver a drug, you may want the carrier to be non-immunogenic. We can tell you exactly which particle you can use for that,” he says. “But if you want to stimulate the immune response, for example to activate the immune system against cancer … then you can use certain particles which will activate the immune response but avoid inflammation. We can produce interferons, but not inflammatory cytokines, for example.
“This is like sharpshooting,” he explains. “You will be shooting for a particular cytokine, without touching others. This is like a letter or a word, like a text message that we send to the immune system. The immune system will read your message and text back with the interferon.”




