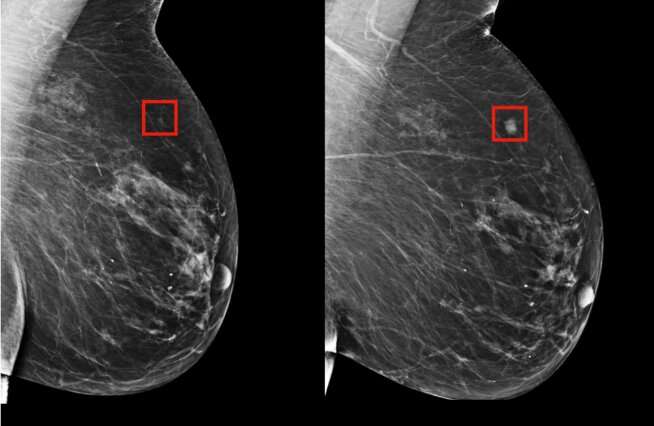
The team’s model was shown to be able to identify a woman at high risk of breast cancer four years (left) before it developed (right). Credit: Massachusetts Institute of Technology
Artificial intelligence (AI) could help doctors predict breast cancer risk earlier and tailor care options to individual patients based on risk.
Researchers from the Massachusetts Institute of Technology’s (MIT) Computer Science and Artificial Intelligence Laboratory (CSAIL) have developed a new technique using a deep-learning model that predicts if a patient is likely to develop breast cancer as much as five years in the future.
The new deep learning algorithm was trained on the 90,000-mammogram results and known outcomes of about 60,000 Massachusetts General Hospital (MGH) patients to learn subtle patterns in breast tissue that act as precursors to malignant tumors.
The researchers are hoping to refine their technique and ultimately use it to allow doctors to customize screening and prevention programs for individuals, eliminating late diagnoses.
“Rather than taking a one-size-fits-all approach, we can personalize screening around a woman’s risk of developing cancer,” MIT professor Regina Barzilay, senior author of the study and a breast cancer survivor, said in a statement. “For example, a doctor might recommend that one group of women get a mammogram every other year, while another higher-risk group might get supplemental MRI screening.”
After testing the model, the researchers found that their method accurately placed 31 percent of all cancer patients in its highest-risk category, while traditional models only predict with 18 percent accurately.
The new deep-learning model was able to detect patterns in mammogram results that were too subtle for the human eye to manually detect.
“Since the 1960s radiologists have noticed that women have unique and widely variable patterns of breast tissue visible on the mammogram,” Constance Lehman, a professor of radiology at Harvard Medical School and division chief of breast imaging at MGH, said in a statement. “These patterns can represent the influence of genetics, hormones, pregnancy, lactation, diet, weight loss, and weight gain. We can now leverage this detailed information to be more precise in our risk assessment at the individual level.”
Another goal for the researchers is to make risk assessment more accurate for racial minorities as current early prediction models are more accurate for white populations than for other races. The new model is equally accurate for all races, which is particularly important for black women who are 42 percent more likely to die from breast cancer for a number of reasons such as differences in detection and a lack of access to health care.
“It’s particularly striking that the model performs equally as well for white and black people, which has not been the case with prior tools,” Allison Kurian, an associate professor of medicine and health research/policy at Stanford University School of Medicine, said in a statement. “If validated and made available for widespread use, this could really improve on our current strategies to estimate risk.”
The information derived from the deep-learning model could also allow doctors to test patients for risks of other diseases and disorders, such as cardiovascular disease or other types of cancer like pancreatic cancer, which does not currently have an accurate risk assessment model.
In the past, there has not been a lot of support in the medical community to conduct risk-based screenings rather than age-based screenings.
“This is because before we did not have accurate risk assessment tools that worked for individual women,” Lehman said. “Our work is the first to show that it’s possible.”
The first breast-cancer risk model was developed in 1989, based on a number of human risk factors like age, family cancer history, hormonal and reproductive factors and breast density.
However, over the last three decades researchers have found that most of those factors only have a weak correlation with breast cancer.
The study was published in Radiology.




