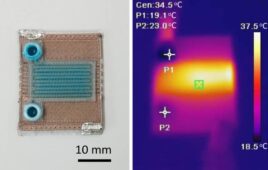
The electronic bandage fits easily into the palm of a hand. [Image courtesy of Northwestern University]
Northwestern University engineering researchers say they developed a first-of-its-kind small, flexible, stretchable electronic bandage.
This bandage accelerates healing by delivering electrotherapy directly to the wound site. It also actively monitors the healing process and harmlessly dissolves — with its electrodes — into the body once no longer needed.
The Northwestern McCormick School of Engineering researchers believe this could become a powerful tool for diabetes patients. Their ulcers can lead to various complications, including amputated limbs or even death. In a study with mice, the bandage healed diabetic ulcers 30% faster than without the bandage.
The researchers published their findings last week in Science Advances. They say it is the first bioresorbable bandage capable of delivering electrotherapy, and the first example of a smart regenerative system.
“When a person develops a wound, the goal is always to close that wound as quickly as possible,” said Guillermo Ameer, who co-led the study. “Otherwise, an open wound is susceptible to infection. And, for people with diabetes, infections are even harder to treat and more dangerous. For these patients, there is a major unmet need for cost-effective solutions that really work for them. Our new bandage is cost-effective, easy to apply, adaptable, comfortable, and efficient at closing wounds to prevent infections and further complications.”
Study co-leader John Rogers said the team designed the bandage with entirely resorbable active components interacting with the wound bed. This means they disappear naturally, avoiding potential damage to the tissue that physical extraction could cause.
Using electricity to power the bandage
Ameer as the Daniel Hale Williams Professor of Biomedical Engineering at the McCormick School of Engineering. He’s also a professor of surgery at Northwestern University Feinberg School of Medicine, among other roles. Rogers, serves as the Louis Simpson and Kimberly Querrey Professor of Materials Science and Engineering, Biomedical Engineering, and Neurological Surgery at McCormick and Feinberg. He also directs the Querrey Simpson Institute for Bioelectronics.
Together, they decided to evaluate if electrical stimulation therapy could help close stubborn diabetic ulcer wounds. This method restores the body’s normal electrical signals after injuries disrupt them, attracting new cells to migrate to the wound bed.
Clinicians have historically used this method for healing. However, this equipment generally includes wired, bulky components that require supervision in a hospital setting. Ameer and Rogers wanted to design a more comfortable product worn around the clock at home.
“Our body relies on electrical signals to function,” Ameer said. “We tried to restore or promote a more normal electrical environment across the wound. We observed that cells rapidly migrated into the wound and regenerated skin tissue in the area. The new skin tissue included new blood vessels, and inflammation was subdued.”
Allowing for remote operation
The bandage Rogers and Ameer developed softly wraps around the injury site. One side contains two electrodes and the other contains an energy-harvesting coil and a near-field communication system for transporting data wirelessly in real time.
Researchers also included sensors that assess how the wound heals, monitoring progress by measuring the resistance of the electrical current across the wound. Gradual decreases reflect directly to the healing process. If physicians observe a high current measurement, they know there is an issue.
These capabilities allow for remote operation without wires. A physician can decide when to apply the electrical stimulation and monitor the healing progress from afar. They applied electrical stimulation for just 30 minutes a day in the animal model study. Even that short amount of time accelerated wound closure by 30%.
“As a wound tries to heal, it produces a moist environment,” Ameer said. “Then, as it heals, it should dry up. Moisture alters the current, so we are able to detect that by tracking electrical resistance in the wound. Then, we can collect that information and transmit it wirelessly. With wound care management, we ideally want the wound to close within a month. If it takes longer, that delay can raise concerns.”
And it’s gone
The researchers developed the electrodes from a metal called molybdenum, commonly used in electronic and semiconductor applications. They discovered that molybdenum can biodegrade when thin enough, and it doesn’t interfere with the healing process.
“We are the first to show that molybdenum can be used as a biodegradable electrode for wound healing,” Ameer said. “After about six months, most of it was gone. And we found there’s very little accumulation in the organs. Nothing out of the ordinary. But the amount of metal we use to make these electrodes is so minimal, we don’t expect it to cause any major issues.”
Next steps include plans to test the bandage for diabetic ulcers in larger animal models. Human tests would ideally follow. The researchers say leveraging the body’s healing power without drugs or biologics means their invention faces fewer regulatory hurdles. They hope this means they could bring their bandage to the market much sooner.
The National Institute of Diabetes and Digestive and Kidney Diseases and CARE partially supported the study. Northwestern biomedical engineering doctoral candidate Joseph Song served as co-first author.