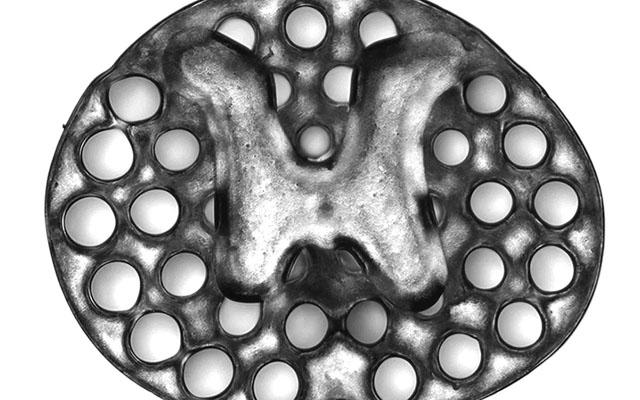
A 3D printed, two-millimeter implant (slightly larger than the thickness of a penny) used as scaffolding to repair spinal cord injuries in rats. The dots surrounding the H-shaped core are hollow portals through which implanted neural stem cells can extend axons into host tissues. Credit: Jacob Koffler and Wei Zhu, UC San Diego
Using new 3D printing technologies, researchers have developed a spinal cord implant that promotes nerve growth in injured sites and restore connections and lost function.
A team from the University of California San Diego School of Medicine and Institute of Engineering in Medicine have for the first time used a rapid 3D printing technique to produce a spinal cord littered with neural stem cells that they successfully implanted into the sites of rats with severe spinal cord injuries.
“In recent years and papers, we’ve progressively moved closer to the goal of abundant, long-distance regeneration of injured axons in spinal cord injury, which is fundamental to any true restoration of physical function,” co-senior author Mark Tuszynski, MD, PhD, a professor of neuroscience and director of the Translational Neuroscience Institute at UC San Diego School of Medicine, said in a statement.
In the rat models, the scaffolds demonstrated tissue regrowth, stem cell survival and the expansion of neural stem cell axons—the long, threadlike extensions on nerve cells that reach out to connect to other cells—out of the scaffolding and into the host spinal cord.
“The new work puts us even closer to real thing because the 3D scaffolding recapitulates the slender, bundled arrays of axons in the spinal cord,” co-first author Kobi Koffler, PhD, assistant project scientist in Tuszynski’s lab, said in a statement. “It helps organize regenerating axons to replicate the anatomy of the pre-injured spinal cord.”
After just a few months, the rats’ spinal cord tissue regrew completely across the injury and connected the severed ends of the host spinal cord. The treated rats regained significant functional motor improvement in their hind legs.
“This marks another key step toward conducting clinical trials to repair spinal cord injuries in people,” Koffler said. “The scaffolding provides a stable, physical structure that supports consistent engraftment and survival of neural stem cells.
“It seems to shield grafted stem cells from the often toxic, inflammatory environment of a spinal cord injury and helps guide axons through the lesion site completely,” he added.
The neural stem cells were also able to survive due to the rats’ circulatory system penetrating the implants to form functioning networks of blood vessels.
The researchers opted for a rapid 3D printing technique that enabled them to produce a scaffold that mimics the central nervous system structures. This technique allowed them to align the axons from one end of the spinal cord injury to the other, while the scaffold keeps them in order to guide them to grow in the right direction to complete the spinal cord connection.
Each implant is comprised of several 200-micrometer-wide channels that guide neural stem cells and axon growth along the length of the injured spinal cord. Using the 3D printing technique, the researchers produce two-millimeter-sized implants in less than two seconds.
The team believe the technique is scalable to human spinal cord sizes and as a proof of concept, they printed, within 10 minutes, four-centimeter-sized implants modeled from MRI scans of injured human spinal cords.
“This shows the flexibility of our 3D printing technology,” co-first author Wei Zhu, PhD, nanoengineering postdoctoral fellow in Chen’s group, said in a statement. “We can quickly print out an implant that’s just right to match the injured site of the host spinal cord regardless of the size and shape.”
To further prove this, the researchers are currently scaling up their technology and testing it on larger animal models. They also plan to incorporate proteins within the spinal cord scaffold that further stimulate stem cell survival and axon outgrowth.
The study was published in Nature Medicine.




