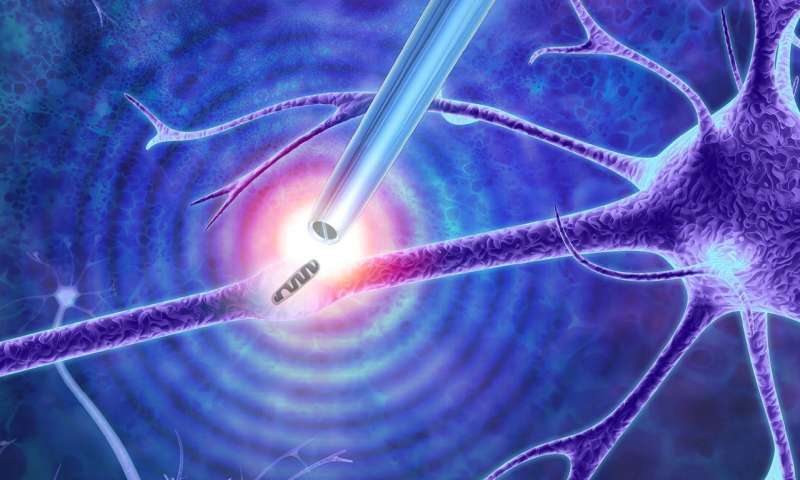
Illustration showing the nanotweezers extracting a mitochondrion from a cell. Credit: Imperial College London
Researchers have begun using nanoscale tweezers that use electrical impulses to extract single DNA, proteins and organelles from living cells without causing damage.
A research team from Imperial College London has developed a new technique based on a phenomenon called dielectrophoresis that enables the tweezers to generate a sufficiently high electric field to enable the trapping of certain objects such as single molecules and particles.
“With our tweezers, we can extract the minimum number of molecules that we need from a cell in real time, without damaging it,” Professor Joshua Edel, from the Department of Chemistry at Imperial, said in a statement. “We have demonstrated that we can manipulate and extract several different parts from different regions of the cell—including mitochondria from the cell body, RNA from different locations in the cytoplasm and even DNA from the nucleus.”
The tweezers used are formed from a sharp glass rod terminating with a pair of electrodes made from a carbon-based material similar to graphite with a tip that less than 50 nanometers in diameter and is split into two electrodes with a 10-to-20-nanometer gap between them.
The gap creates a powerful and highly localized electrical field that can trap and extract the small contents of cells, such as DNA and transcription factors, when an alternating current voltage is applied.
The new technique could be potentially used to carry out experiments not currently feasible.
“These nanoscale tweezers could be a vital addition to the toolbox for manipulating single cells and their parts,” Alex Ivanov, PhD, from the Department of Chemistry at Imperial, said in a statement. “By studying living cells at the molecular level, we can extract individual molecules from the same location with unprecedented spatial resolution and over multiple points in time.
“This may provide a deeper understanding of cellular processes, and in establishing why cells from the same type can be very different to each other,” he added.
For example, nerve cells require much energy to fire messages around the body, so they contain many mitochondria to help them function. However, by adding or removing mitochondria from individual nerve cells, researchers could better understand their role, especially in neurodegenerative diseases.
It is difficult to gain a true understanding of how cells function, particularly for individual cells that are of the same type but have very different compositions at the single-molecule level like the brain, muscles and fat cells.
By cataloguing, the diversity of seemingly identical cells researchers could better understand fundamental cellular processes and design better models of disease and new patient-specific therapies.
Traditional methods to study these differences usually involve bursting the cell, resulting in all of its contents getting mixed, resulting in both a loss of spatial information—like how the contents were laid out in relation to each other and dynamic information, such as the molecular changes in a cell over time.
The study was published in Nature Nanotechnology.




