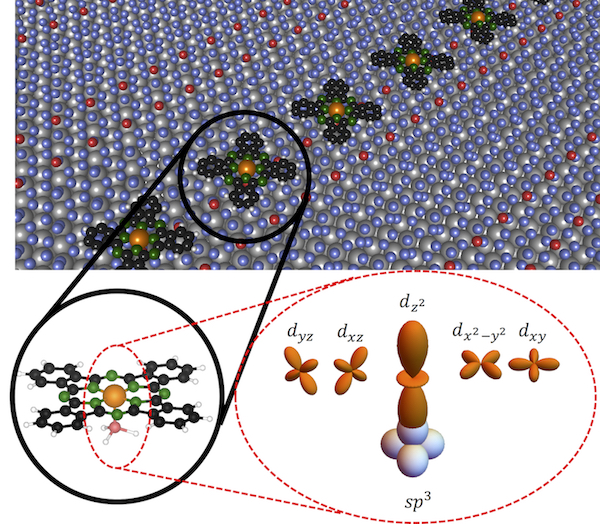
Top shows a ball model of the silicene surface on ZrB2 with iron phthalocyanine molecules arranged along the silicene domain boundaries. The bottom left close-up shows the bonding of the molecule to one of the silicon atoms in the domain boundary, while the bottom right close-up highlights the fact that only molecular orbitals perpendicular to the surface are involved in the bonding. This leaves the molecular states that are composed from the remaining in-plane orbitals largely unaffected. Image: Courtesy of Tobias G. Gill, Vasile Caciuc, Nicolae Atodiresei, and Cyrus Hirjibehedin
A new way to control the positioning of individual molecules on the surface of materials that are only a single layer of atoms thick has been discovered by a team of scientists from the LCN at UCL in the U.K.; the Japan Advanced Institute of Science and Technology (JAIST) and the Institute for Solid State Physics at the University of Tokyo in Japan; and the Forschungszentrum Jülich in Germany.
Reporting their results in the journal Advanced Materials, the team describes how the bonding between an iron phthalocyanine (FePc) molecule — which resembles haem in blood — and a single layer of silicon can be strong enough to pin individual molecules at room temperature without causing large distortions in the molecule’s electronic properties.
Over the last decade, there has been an ever-expanding list of materials whose thickness is only a few atomic layers. These so-called “two-dimensional” (2D) materials display many exciting new properties by themselves, but their range of functionality can be further expanded by creating precise arrangements of molecules on top. However, many 2D materials have only weak interactions with molecules deposited on top of them. While this prevents detrimental modification of important molecular properties, it also makes it difficult to fix the position of molecules, especially at room temperature.
One way around this problem is to modify the structure of the 2D material by placing it on the surface of another material. The resulting mismatch between the two materials can cause the 2D material to restructure, creating some areas on the surface that are different from others. This is particularly true for the 2D material silicene, which is composed of a single layer of silicon atoms. “The interactions between silicene and the zirconium diboride (ZrB2) crystal that it forms on causes it to form striped domains with interesting domain boundaries in between,” says Yukiko Yamada-Takamura, whose group first developed the technique for forming silicene on ZrB2.
Using this nanostructured silicene formed on ZrB2, the scientists found that FePc molecules preferentially bond to only the nanoscale linear boundaries between different domains of silicene. This was done by imaging the molecules on the surface of silicene with atomic resolution using a scanning tunneling microscope. With this technique, the scientists were able to show that the molecules’ electronic properties closely resembled those of an isolated molecule even though the molecules were strongly bound to the silicene surface.
To understand the unique nature of the bonding, and why it allowed the molecules to retain their important properties, the team performed complex density functional theory calculations. This allowed them to understand both the electronic properties of the silicene surface and how these might interact with a molecule that was placed on top of the silicene domain boundaries.
“Our calculations revealed that metal atoms in planar molecules such as iron phthalocyanine can form additional bonds with localized orbitals that extend out from the silicon atoms at these domain boundaries, strongly fixing the position of the molecules such that they are stable even at room temperature,” says Nicolae Atodiresei, who coordinated the theoretical calculations. “Because a distinct set of molecular orbitals that are oriented perpendicular to the 2D surface is involved in binding the molecule, the in-plane molecular states are not strongly affected.”
Although most of the measurements were performed at very low temperatures (a few degrees above absolute zero), the team also demonstrated that the molecules remained pinned even up to room temperature, making the results more likely to have practical implications. “These results highlight that nanostructured 2D materials can provides unique interactions for enabling new ways to control the assembly of functional molecules,” explains Cyrus Hirjibehedin, the project’s lead scientist. “This could lead to new applications, such as providing new ways to transmit and process information at the single molecule level.”
Source: London Centre for Nanotechnology




