The new method for designing solid-state batteries starts with liquid electrolytes inside the electrochemical cell. Special molecules then initiate polymerization, improving contact between the electrolyte and electrodes. Credit: Qing Zhao
While it has long been known that solid-state batteries are a safer and more energy dense alternative to the lithium-ion batteries commonly used for electric vehicles and personal electronics, challenges remain that prevent them from being implemented on a wider-scale.
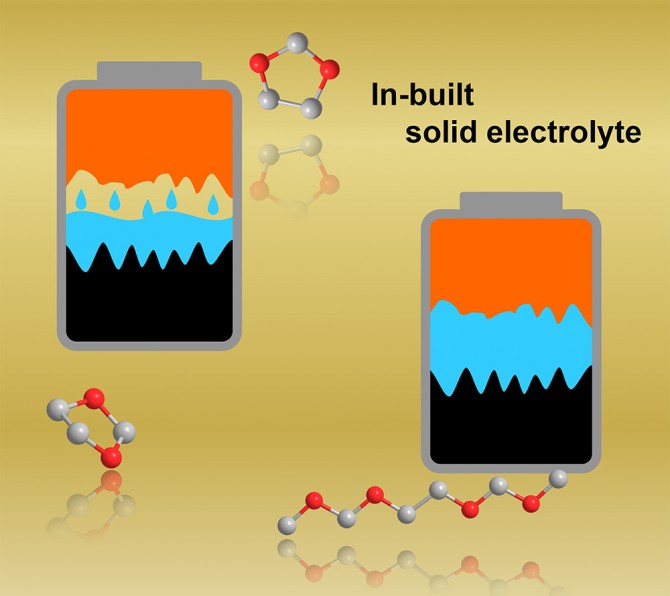
However, a research team from Cornell University has discovered that by starting with liquid electrolytes that are then transformed into solid polymers inside of an electrochemical cell, they can obtain the benefits of both liquid and solid properties, preventing some of the limitations of current solid-state battery designs.
“Imagine a glass full of ice cubes: Some of the ice will contact the glass, but there are gaps,” Qing Zhao, a postdoctoral researcher and lead author on the study, said in a statement. “But if you fill the glass with water and freeze it, the interfaces will be fully coated, and you establish a strong connection between the solid surface of the glass and its liquid contents. This same general concept in a battery facilitates high rates of ion transfer across the solid surfaces of a battery electrode to an electrolyte without needing a combustible liquid to operate.”
Some of the current limitations preventing solid-state batteries from more widespread usage include high manufacturing costs and poor interfacial properties that present significant technical hurdles.
To overcome these issues, the researchers used special molecules that can initiate polymerization inside of the electrochemical cell without compromising the other functions of the cell. If the electrolyte is a cyclic ether, the initiator can be designed to rip open the ring and produce reactive monomer strands that bond together to create long chain-like molecules with essentially the same chemistry as the ether. The solid-polymer will now retain the tight connections at the metal interfaces.
The solid-state batteries can also enable next-generation batteries to better utilize metals such as lithium and aluminum as anodes for achieving far more energy storage than what today’s state-of-the-art batteries are capable of. The solid-state electrolyte will prevent these metals from forming dendrites—short strands of lithium that grow inside of batteries that could potentially cause them to short circuit, leading to overheating and failure.
Solid-state batteries do circumvent the need for battery cooling because they provide stability to thermal changes.
“Our findings open an entirely new pathway to create practical solid-state batteries that can be used in a range of applications,” senior author Lynden Archer, the James A. Friend Family Distinguished Professor of Engineering in the Smith School of Chemical and Biomolecular Engineering, said in a statement.
Archer said that the new strategy also could lead to extending battery life cycle and recharging capabilities of high-energy-density rechargeable metal batteries.
The study was published in Nature Energy.




