Scientists have a better understanding of the immune system at a molecular level, thanks to University of Queensland-led research that may now lead to a range of new treatments for disease.
The research provides a new foundation for therapeutic strategies against a wide range of diseases and infections, said Professor Bostjan Kobe.
“This study shows for the first time that signalling happens downstream of ‘toll-like receptors’ – proteins that play a key role in the innate immune system,” Professor Kobe said.
“These receptors are a double-edged sword,” he said. “On one hand, they protect us against infection, but they are also responsible for pathological inflammatory responses in a range of disorders from chronic inflammatory, autoimmune, cardiovascular and cancer-related disorders to pathologies associated with infections. For this reason, the associated pathways are important drug targets for a range of diseases.”
Professor Kobe, of the School of Chemistry and Molecular Biosciences and the Australian Infectious Diseases Research Centre, said the signalling assemblies’ structure provided the means to design therapeutic drugs using rational, structure-based approaches.
“These are similar to approaches Australian researchers used to develop the anti-flu drug Relenza,” he said.
Innate immune receptors, particularly toll-like receptors, were discovered only in recent years, Professor Kobe said, with the 2011 Nobel Prize in Physiology or Medicine co-awarded to Bruce Beutler and Jules Hoffmann for their discovery.
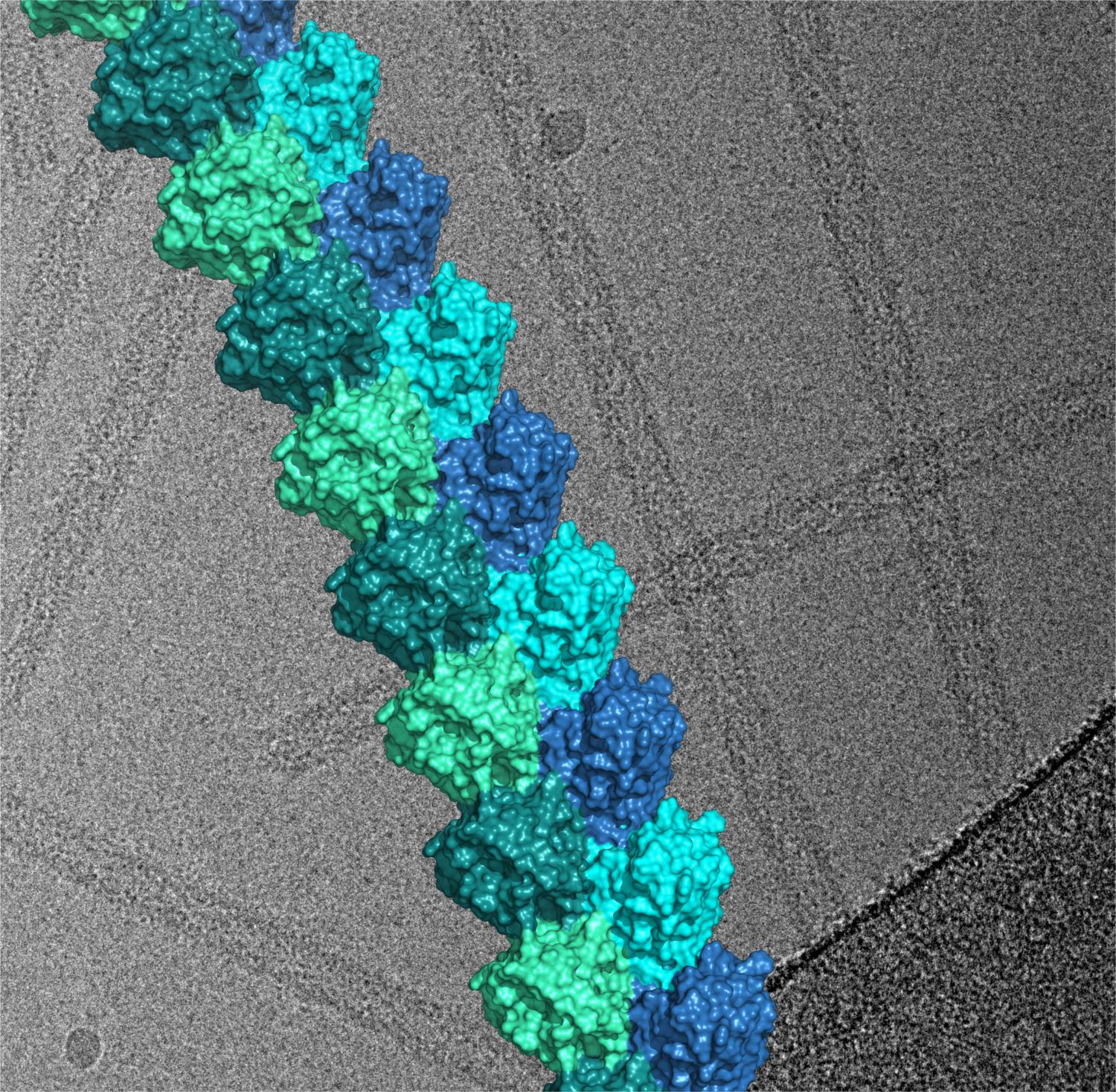
“The mechanism of signalling has been a matter of controversy,” he said. “Many incorrect molecular models were proposed over last 15 years, but the data they were based on was too limited. We managed to reconstitute the signalling assembly, determine its structure by using cryo-electron microscopy, and provide conclusive evidence that explains signalling in the cell.”
He added: “This is a rather novel type of signalling we have called SCAF (Signalling by Cooperative Assembly Formation). A large assembly forms very quickly after minimal stimulus, achieving very high amplification very rapidly. This is exactly what’s needed in the first stage of immune response.”
In a report published in the journal Nature Structural and Molecular Biology (DOI tba), the international research team described how the signal is amplified after the membrane receptor senses a pathogen and initiates an inflammatory response.
A National Health and Medical Research Council project grant funded the study.
The international collaborators included the teams of Dr Thomas Ve who started work in Professor Kobe’s lab at UQ and recently moved to Griffith University, Dr Kate Stacey, Dr Michael Landsberg and Dr Mehdi Mobli at UQ, Dr Yann Gambin and Dr Emma Sierecki at the University of New South Wales, and Professor Ed Egelman at the University of Virginia, USA.




