 Sensors to detect viruses, explosives or drugs often use a technique called surface-enhanced Raman spectroscopy (SERS) in which light wavelengths from a chemical or biological species adsorbed on metal nanostructures are detected and mapped to the identity of the species. In SERS, detection efficiency depends on the surface morphology of the nanostructure. Now, scientists have developed a simple method that could enable the low-cost fabrication of stable and high-performance polymer-based nanostructures at scale.
Sensors to detect viruses, explosives or drugs often use a technique called surface-enhanced Raman spectroscopy (SERS) in which light wavelengths from a chemical or biological species adsorbed on metal nanostructures are detected and mapped to the identity of the species. In SERS, detection efficiency depends on the surface morphology of the nanostructure. Now, scientists have developed a simple method that could enable the low-cost fabrication of stable and high-performance polymer-based nanostructures at scale.
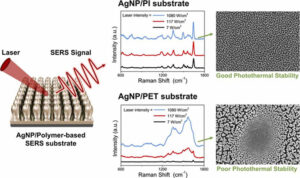
From identifying of viruses, to drugs, to explosives, the surface-enhanced Raman spectroscopy (SERS) technique can do it all and with great sensitivity and accuracy. This is why it is widely used in industry. In SERS, light is shone on a nanosurface containing a chemical or biological analyte. The light reflected after interaction with the analyte then hits a detector which maps the received signal to known compounds to identify the analyte. The interaction of the light with the nanosurface enhances its scattering from the analyte molecules. Thus, the strength of the signals depends on the morphology of the substrate. But developing effective nanosurfaces is, at present, quite challenging, involving complex and expensive processes and difficulty in obtaining materials that can withstand the high heat and light conditions needed for fabrication.
Now, scientists from Pusan National University, Korea, have developed a method — using a heat-resistant polymer called polyimide (PI) — that can easily and rapidly yield SERS nanosurfaces with superior stability and performance at low cost. Their findings are published in the Journal Sensors and Actuators B: Chemical.
Their method comprises a simple two-step process and is carried out in a vacuum chamber with custom-built sputtering apparatus. First the surface of a PI nanofilm is etched via plasma etching in the presence of argon ions to form a nanotextured surface consisting of nanopillars. The PI nanopillars are then uniformly coated with silver (Ag) nanoparticles (NPs) through a standard deposition process. The vacuum prevents any external contamination, yielding highly uniform AgNP/PI nanostructures ready as platforms for analytes.
By controlling the time taken from PI etching to AgNP deposition, the scientists were able to control both the nanopillar density and the Ag coating thickness to arrive at a platform with the best morphology for an optimal signal intensity. They even tested the performance of this platform experimentally, for the detection of a chemical called RhB, and found it reliable.
“Our research shows that it is possible to fabricate a sensor with superior performance at a lower cost than current noble-metal-based SERS sensors,“ said Prof. Young-Rae Cho, who led the study. Further, the scientists expect this fabrication process to significantly expand the scope of applicability of the SERS technique. “Our method could facilitate the dissemination of disease diagnosis kits in households and enable even more widespread use of excellent detection sensors in the virus, explosives and drug detection industries,” said Prof. Cho, looking towards a much more hopeful future.





Tell Us What You Think!