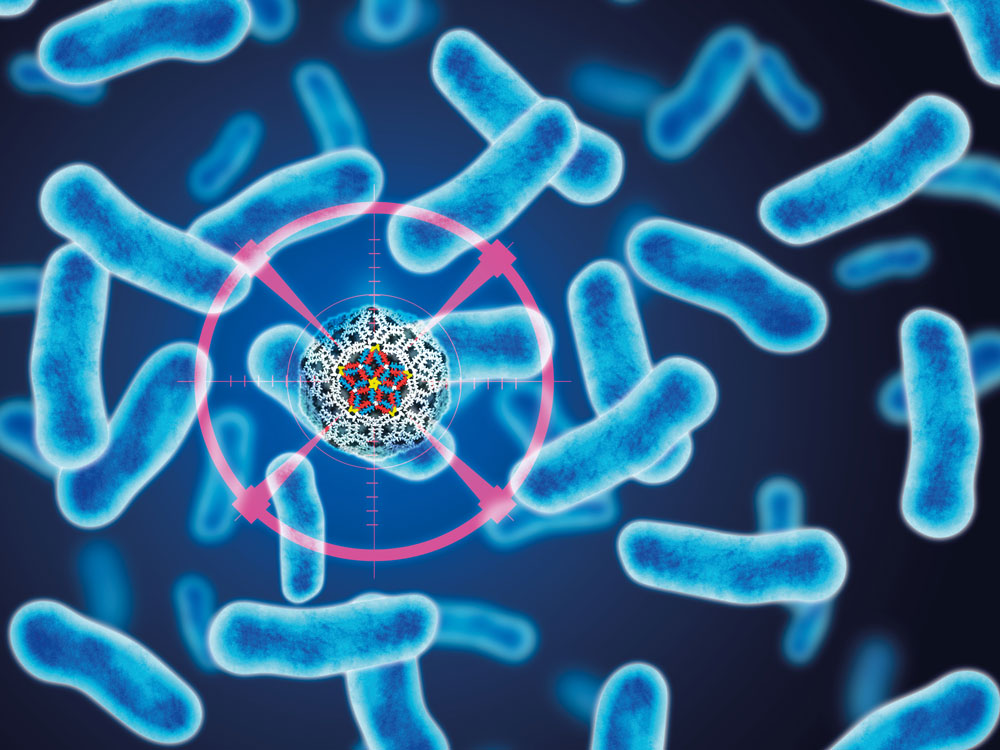
A synthetic “virus” that kills bacteria on first contact has been developed by University College London (UCL) and National Physical Laboratory researchers (NPL).
The study, published in Nature Communications, shows how newly designed proteins can be used to build tiny hollow shells that emulate the outer structures of naturally occurring viruses. The synthetic virus “drones” recognize bacterial cells before targeting and destroying their most vulnerable part — their membrane.
“We used high-resolution and real-time imaging to see the impact of the synthetic viruses on bacterial model membranes and found that they are extremely destructive,” explains co-author and UCL EngD student Hasan Alkassem (NPL, UCL London Centre for Nanotechnology, UCL Biochemical Engineering, and EPSRC Centre for Doctoral Training in Bioprocess Engineering Leadership).
“Seconds after landing on the surface, the synthetic viruses disassemble and form rapidly expanding holes in the membrane, causing it to leak. Experiments on intact bacteria then showed that this caused the bacteria to die.”
The discovery is important as it demonstrates a possible new approach to tackling the ever-growing issue of antibiotic resistance, by opening new options for the treatment of infectious diseases.
More than 700,000 people across the world die from drug resistant infections every year and antibiotic discovery has fallen well behind its historical rate, with traditional discovery methods being exhausted.
“The way our synthetic virus works means that bacteria are less likely to become resistant to it. They also have an additional advantage over traditional antibiotics in that they don’t have to reach and hit a single target inside a bacterial cell to be effective,” says co-author and UCL PhD student, Alex Yon (UCL London Centre for Nanotechnology and UCL Institute of Structural & Molecular Biology).
Furthermore, because such synthetic viruses leave human cells unaffected, but have the ability to enter them like natural viruses do, they hold promise for gene delivery and gene editing — core capabilities for gene therapy and synthetic biology — as well as for killing bacteria that hide inside human cells.
Lead author, Dr. Max Ryadnov, the NPL’s science leader in biometrology, explains, “This discovery adds to the growing toolbox of engineering metrology methods and materials being developed at NPL to realize the full potential of synthetic biology for industry and healthcare. This research also offers long-term and creative solutions for alternative treatments of infectious diseases that are urgently needed.”
Professor Bart Hoogenboom (UCL London Centre for Nanotechnology, UCL Physics & Astronomy and UCL Institute of Structural & Molecular Biology), who supervised the UCL part of this research, concludes: “We were particularly pleased to find that our observations of membrane damage at nanometer-length scale translated into the actual bacteria being killed. Hence we have a potential strategy to treat infectious diseases, and we also understand how it works.”
The work was funded by the Engineering and Physical Sciences Research Council (EPSRC), European Metrology Programme for Innovation and Research (EMPIR), the Department for Business, Innovation and Skills (BEIS), and the Biotechnology and Biological Sciences Research Council (BBSRC). Specialist measurements were performed at the Diamond Light Source.
Source: University College London




