Advanced Microscope Technology Reveals Novel Side to Cell Signaling Complex
In a study published today in Science, UT Southwestern and Rockefeller University researchers used advanced microscopes to determine at atomic resolution the structure of a molecular complex implicated in birth defects and several cancers.
The Hedgehog signaling pathway, which transmits information to embryonic cells, is crucial to human health. Insufficient signaling during development leads to birth defects, while unrestrained Hedgehog signaling occurs in many cancers. Excessive signaling is implicated in basal cell carcinoma – the most common malignant cancer in humans – as well as in brain cancer, breast cancer, and prostate cancer, said Dr. Xiaochun Li, UT Southwestern Assistant Professor of Molecular Genetics and Biophysics and a Rita C. and William P. Clements, Jr. Scholar in Biomedical Research. Many pharmaceutical companies are developing drugs that target Hedgehog signaling. Having a clearer view of the structure could help those efforts, Dr. Li said.
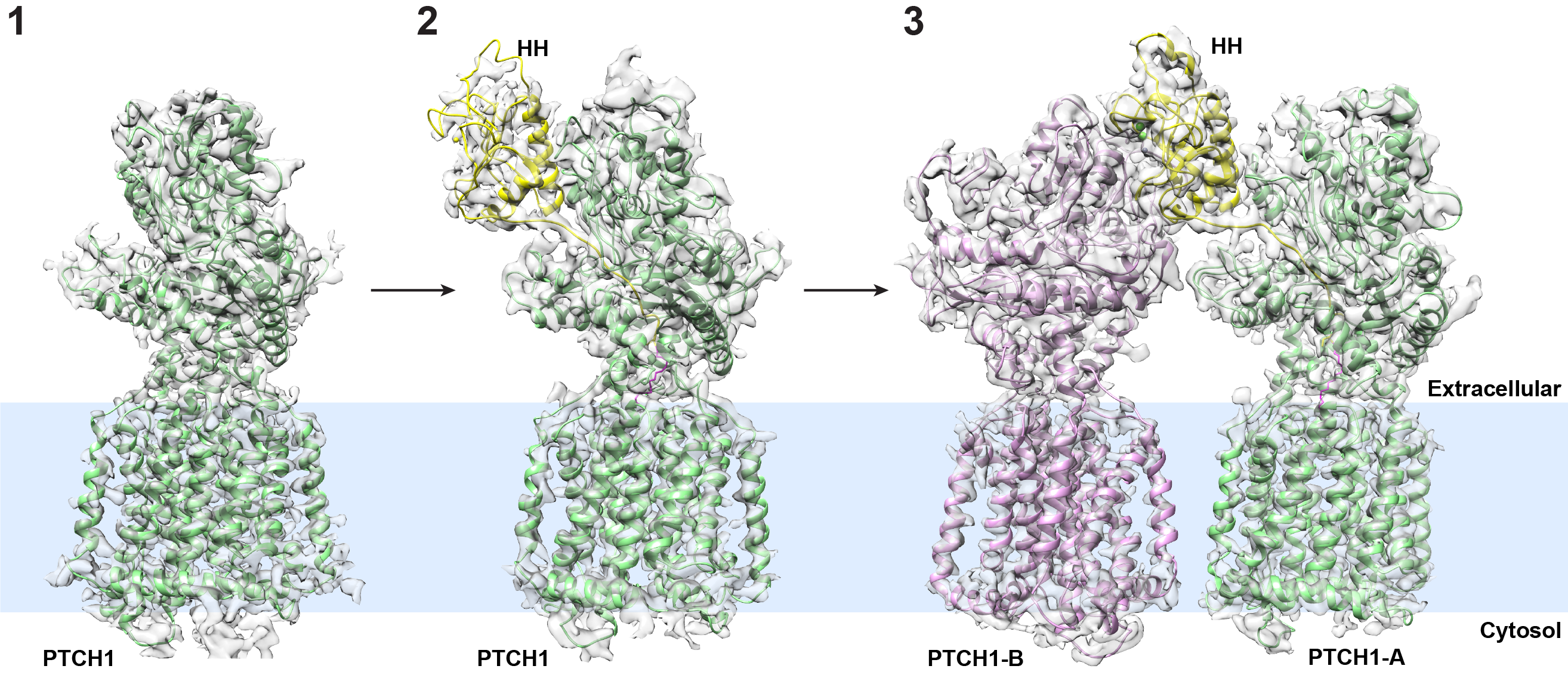
The researchers, using cryo-electron microscopy (cryo-EM) technology, showed that two Patched-1 (PTCH1) molecules simultaneously engage a single Hedgehog (HH) molecule, but at two distinct sites. This unique 2-to-1 ratio PTCH1-HH complex is required for efficient Hedgehog signaling in cells.
Cryo-EM uses enormous microscopes equipped with robotics to determine the structure of molecular samples that are frozen at temperatures so low that ice crystals cannot form.
In another paper published in Nature last month, Dr. Li’s group reported a cryo-EM structure of the 1-to-1 PTCH1-HH complex. Their biochemical assays indicated that HH binding to one PTCH1 molecule may not be sufficient for full activity. HH may need to recruit either a different protein or another PTCH1 molecule, he said.
“In the current Science paper, we report a 2-to-1 PTCH1-HH complex in which one Hedgehog molecule binds to two of its receptors (PTCH1) at two different spots. We used our cell biology assay to verify that this 2-to-1 complex is indeed the signaling generator for Hedgehog signaling. Combined with the earlier study published in Nature, we hope our new work will provide additional insights for physicians and scientists in this field,” he explained.
Co-authors include Molecular Genetics postdoctoral researcher Dr. Xiaofeng Qi and research assistant Philip Schmiege from UT Southwestern and Dr. Elias Coutavas of Rockefeller University.
The study received support from the Cancer Prevention and Research Institute of Texas (CPRIT), the UT Southwestern Endowed Scholars Program in Medical Science, the O’Donnell Junior Faculty Funds, The Welch Foundation, the National Institutes of Health, and Rockefeller University.
The authors dedicated the study to the late Dr. Günter Blobel, 1999 Nobel Laureate in Physiology or Medicine, with whom Dr. Li determined the structure of the Niemann-Pick C1 (NPC1) protein when Dr. Li was a postdoctoral researcher in the Blobel laboratory at Rockefeller University. The structures of the NPC1 and the newly elucidated PTCH1 share some structural similarities.




