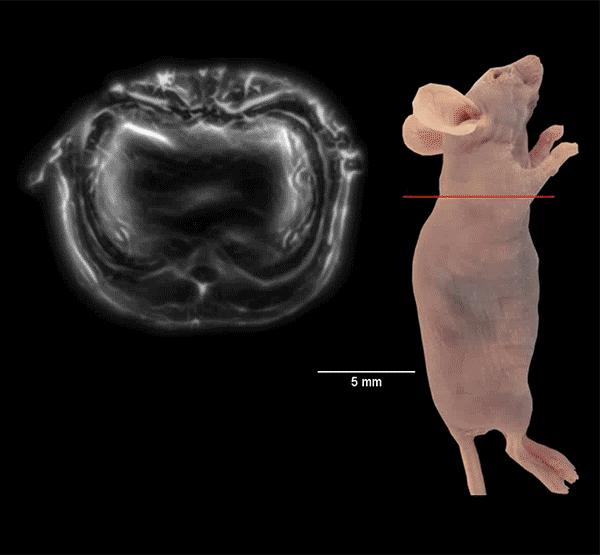
Photoacoustic imaging uses light to induce cells to emit ultrasound waves. Advances in the technology now allow it to scan entire cross-sections of a live mouse in real time for an entire body view in under a minute. Source: Junjie Yao & Kara Manke, Duke University
Biomedical engineers are now able to take a live, holistic look at the inner workings of a small animal with enough resolution to see active organs, flowing blood, circulating melanoma cells and firing neural networks.
The technique dubbed “single-impulse photoacoustic computed tomography (SIP-PACT)” uses the best of both light and ultrasound to peer inside living animals. Researchers at Duke University and CalTech have shown this hybrid imaging technology breaks the longstanding resolution and speed barriers in small-animal whole-body imaging. It provides a full cross-sectional view of a small animal’s internal functions in real time.
The results appear online on May 10, 2017, in Nature Biomedical Engineering.
“Photoacoustic imaging has been highly expected to get real-time whole-body imaging of a small animal with rich functional information,” said Junjie Yao, assistant professor of biomedical engineering at Duke University. “With this advance, researchers can easily watch as drugs are distributed throughout an animal and track how different organs respond.”
Photoacoustic imaging combines a variety of imaging techniques into one platform.
Traditional light-based microscopy provides fast, high-resolution images that retain important functional information based on the wavelengths of light (i.e., colors) that the tissue absorbs, reflects or emits. The significant amount of light that scatters as it travels through tissue, however, limits the depth of light microscopy to just a few millimeters.
Ultrasound waves easily travel through tissue, providing a much more in-depth view, but do not have the ability to read tissue’s chemical components and miss much of the important information that light carries with it. Magnetic resonance imaging (MRI) can also see deep into tissue, but requires a strong magnetic field and often takes seconds to minutes to form an image. X-rays and positron emission tomography (PET) deliver too much radiation to the subject to be practical over long time periods.
Photoacoustic imaging uses powerful but extremely short laser bursts that safely cause cells to emit ultrasound waves, which then travel unimpeded back through the tissue.
“It’s basically compressing one second’s worth of summer-noon sunlight over a finger nail area into a single nanosecond,” said Yao, who has been working with the technology for nearly a decade. “When the laser hits a cell, the energy causes it to heat up a tiny bit and expand instantaneously, creating an ultrasonic wave. It’s like the difference between pushing on something to slowly move it and striking it to cause a vibration.”
The result is an imaging technique that can peer up to five centimeters into the typical biological tissue with sub-millimeter-level resolution while retaining the functional information provided by traditional optical microscopy. For example, melanin absorbs near-infrared light, while blood’s reaction to light differs depending on how much oxygen it is carrying.
In the new paper, Yao and colleagues led by Dr. Lihong Wang at the California Institute of Technology add the highly desired speed and panoramic views to the imaging technology’s repertoire. They have built a circular ultrasonic detector and a fast data-acquisition system that can triangulate the origin of an ultrasonic wave from anywhere within the body of a small animal. And with the help of a fast laser that operates within the safety limit, the upgraded device can image the full cross-section of an adult rat 50 times per second, providing detailed movies of its inner workings with 120-micrometer resolution.
“The panoramic effect provides information from all directions and all angles, so you do not lose any information from each laser shot,” said Yao. “You can see the dynamics of the body in action — the pumping of the heart, the dilation of arteries, the functioning of various tissues.”
In the paper, Yao and colleagues describe how they use these abilities to track cancerous melanoma cells traveling through blood vessels of a mouse. They also demonstrate the ability to watch entire neural networks firing in real time.
“This approach is especially powerful because it does not rely on the injection of any type of contrast agent,” said Yao. “You can be sure that changes are not caused by foreign variables. We think that this technology holds great potential for both pre-clinical imaging and clinical translation.”