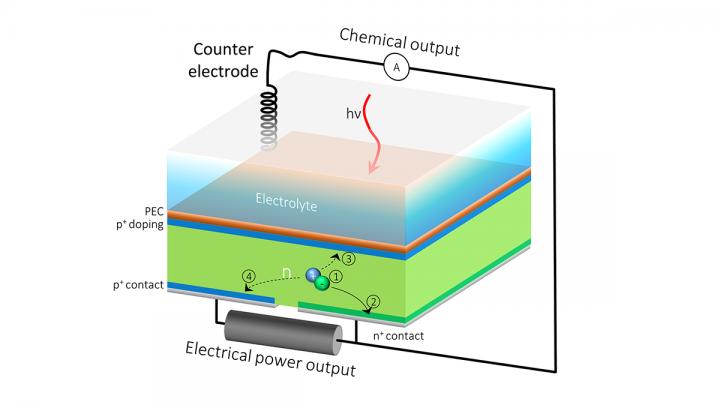
The HPEV cell’s extra back outlet allows the current to be split into two, so that one part of the current contributes to solar fuels generation, and the rest can be extracted as electrical power. Credit: Berkeley Lab, JCAP
Scientists have developed a water-splitting device that is able to generate two different types of energy, while bypassing some of the limitations from current artificial photosynthesis devices.
A research team from the U.S. Department of Energy’s Lawrence Berkeley National Laboratory and the Joint Center for Artificial Photosynthesis has developed a new device called a hybrid photoelectrochemical and voltaic (HPEV) cell that converts sunlight and water into hydrogen fuel and electricity.
Water splitting is an artificial photosynthesis technique where sunlight is used to generate hydrogen fuel from water. However, there previously has not been a design for materials with the right combination of optical, electronic and chemical properties required for them to work efficiently.
The majority of water-splitting devices are made of a stack of light-absorbing materials, where each layer absorbs different wavelengths of the solar spectrum, ranging from less-energetic wavelengths of infrared light to more energetic wavelengths of visible or ultraviolet light.
Each layer builds an electrical voltage when it absorbs light that combine into one voltage large enough to split water into oxygen and hydrogen fuel.
However, the potential for high-performance is compromised in this configuration when they are part of a water-splitting device. Other materials in the stack that do not perform as well as silicon limit the current passing through the device and the system produces much less current than it potentially could, resulting in less solar fuel produced.
“It’s like always running a car in first gear,” Gideon Segev, a postdoctoral researcher at JCAP in Berkeley Lab’s Chemical Sciences Division and the study’s lead author, said in a statement. “This is energy that you could harvest, but because silicon isn’t acting at its maximum power point, most of the excited electrons in the silicon have nowhere to go, so they lose their energy before they are utilized to do useful work.”
In water-splitting devices, the front surface is generally dedicated to solar fuel production, with the back surface serving as an electrical outlet. In their new device, the researchers added an additional electrical contact to the silicon component’s back surface, producing a device with two contacts in the back rather than one.
The extra back outlet allows the current to be split into two, so that one part of the current contributes to solar fuel generation and the other part can be extracted as electrical power.
“And to our surprise, it worked!” Segev said. “In science, you’re never really sure if everything’s going to work even if your computer simulations say they will. But that’s also what makes it fun. It was great to see our experiments validate our simulations’ predictions.”
Based on their calculations, a conventional solar hydrogen generator that is comprised of a combination of silicon and bismuth vanadate would generate hydrogen at a solar to hydrogen efficiency of 6.8 percent.
The HPEV cells also harvest leftover electrons that do not contribute to fuel generation, but rather are used to generate electrical power. This results in a substantial increase in the overall solar energy conversion efficiency.
The researchers will now examine whether they can use the HPEV concept for other applications, including reducing carbon dioxide emissions.
“This was truly a group effort where people with a lot of experience were able to contribute,” Segev said. “After a year and a half of working together on a pretty tedious process, it was great to see our experiments finally come together.”
The study was published in Nature Materials.




