Researchers at Nanjing University published a new study in Environmental and Biogeochemical Processes describing a method for treating perfluorooctanoic acid (PFOA) in water. By introducing a small amount of formic acid into a UV-activated persulfate system, the researchers increased defluorination from 27% to 89% in 24 hours. The best results required acidic conditions (pH 2.5), an oxygen-free environment and 24 hours of 254-nm UV exposure. The method generates carbon dioxide radical anions, which are capable of attacking PFOA’s stable carbon-fluorine bonds.
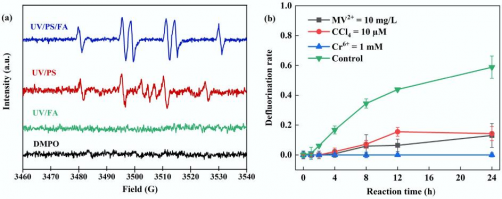
(a) EPR analysis of UV/PS/FA, UV/PS, and UV/FA systems. (b) Free radical quenching experiments. Reaction conditions: PFOA = 20 µM, initial pH of 2.5, anaerobic environment. (a) PS = 2 mM, FA = 15 mM, DMPO = 100 mM, reaction time = 5 min, anaerobic environment. (b) PS = 4 mM, FA = 2 mM, MV2+ = 10 mg/L, CCl4 = 10 µM, Cr(VI) = 1 mM. Credit: Chinese Academy of Sciences
PFOA is part of a class of per- and polyfluoroalkyl substances, called PFAS, that are widely used in textiles, firefighting foams, packaging, electroplating and other industrial applications. Due to their strong carbon-fluorine bonds, the substances persist in the environment and accumulate in organisms, posing risks to liver function, immune systems and reproduction.
Separation technologies such as ion exchange resin (IXR) and granular activated carbon (GAC) can remove PFAS from water; however, the contaminants remain in the environment and continue to cause health risks. IXR results in a brine solution that contains high concentrations of PFAS, which must be disposed of through thermal treatment, underground injection or secure storage.
PFOA contains extremely strong carbon-fluorine bonds that make it difficult to break down. Traditional UV and oxidation methods are often inefficient at destroying the compounds unless conditions are extreme.
The new study showed that introducing formic acid to a UV-activated persulfate system increased defluorination. In other UV methods, the light activates the persulfate to create oxidizing radicals, which tend to attack the carboxyl end of PFOA rather than the carbon-fluorine backbone.
When formic acid is added, the system becomes reductive. The oxidizing radicals react with the acid to form carbon dioxide radical anions, strongly reductive compounds that can attack the carbon-fluorine bonds. Both persulfate and formic acid are inexpensive commodity chemicals, though the study did not include a full cost analysis for scaled-up treatment.
Unlike IXR, the new method does not produce a PFAS-heavy waste brine. However, the degradation process does generate short-chain perfluorocarboxylic acids as intermediates. When PFOA concentrations increased from 20 to 200 µM, defluorination efficiency dropped from roughly 85% to about 55%. The method could potentially be combined with concentration technologies like IXR to concentrate PFAS before destruction.