Supplementing a single protein found in the spinal cord could help prevent symptoms of Lou Gehrig’s disease, according to a new study out of Case Western Reserve University School of Medicine. Researchers found high levels of the protein–called mitofusion 2 or Mfn2–prevented nerve degeneration, muscle atrophy, and paralysis in a mouse model of the disease. Since Mfn2 is often depleted during Lou Gehrig’s, the new study suggests supplementing it could be a novel therapeutic approach for the disease.
Lou Gehrig’s disease, or amyloid lateral sclerosis (ALS), is a progressive disorder that devastates motor nerve cells. People with ALS slowly lose the ability to control muscle movement, and are ultimately unable to speak, eat, move, or breathe. The cellular mechanisms behind ALS are also found in certain types of dementia. For the estimated 15,000 Americans living with ALS, the findings offer new hope for ways to delay symptoms.
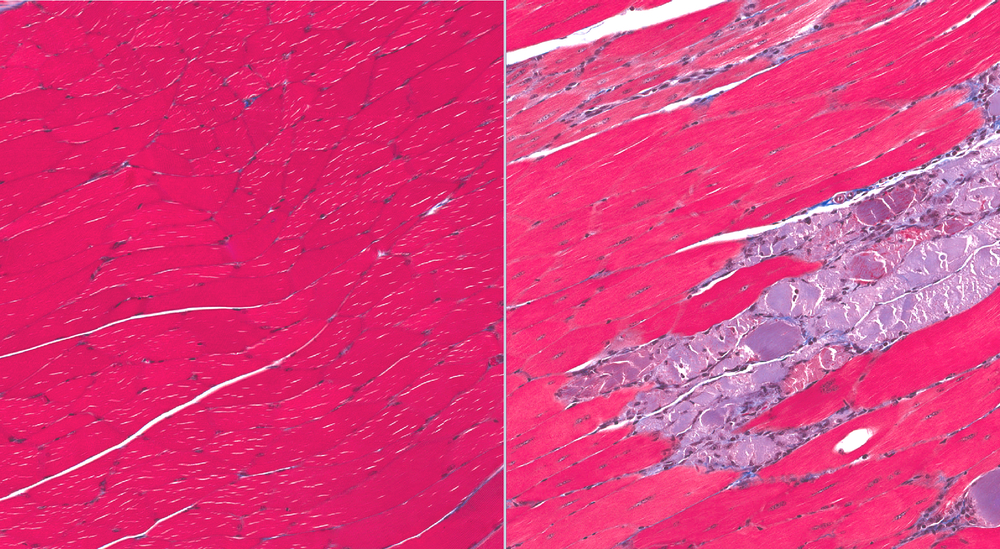
“We found a way to alleviate age and ALS-related muscular atrophy in our mouse models,” said Xinglong Wang, PhD, associate professor of pathology at Case Western Reserve University School of Medicine. “Amazingly, we could delay ALS symptom onset by 67 days.”
Wang led the study, published today in Cell Metabolism, in which researchers successfully staved off muscle atrophy and paralysis simply by increasing Mfn2 levels in mouse spinal cords.
Wang and colleagues tested the most widely used ALS mouse model. They genetically engineered the diseased mice to have increased Mfn2 levels–but only in nerve cells that extend from the spinal cord and connect to muscle fibers. In late stages of the disease, mice with high Mfn2 levels in these nerves were a healthy weight, and did not have any of the muscle atrophy, gait abnormalities, or reduced grip strength that mice in control groups developed. Even mice who underwent heavy sciatic nerve damage benefited from elevated Mfn2 levels.
Said Wang, “Upregulation of Mfn2 specifically in nerve cells is sufficient to abolish skeletal muscle loss in ALS and aged mice, despite ALS-causing protein being found in all organs and tissues.”
By studying nerve cells collected from the mice, Wang’s team uncovered how Mfn2 offers its protective effects. The researchers found Mfn2 coexists with nutrients in cell structures called mitochondria. Their experiments showed mitochondria travel along nerve cell extensions–axons–and deliver the nutrients to the point where nerve cells and muscle fibers meet. This preserves sensitive connections–synapses–between nerve and muscle cells and prevents muscle atrophy. “We found mitochondria function as miniature ‘trucks’ to transport protein along axons to prevent synaptic degeneration,” explained Wang.
Cellular transport is not typically in the job description for mitochondria. The ancient cellular structures are well-known to be “powerhouses of the cell”–producing energy that keeps cells running. According to Wang, “this is a novel, previously unrecognized role for mitochondria.”
Specifically, Wang’s team found mitochondria use Mfn2 on their surfaces to carry a nutrient called calpstatin. Calpstatin inhibits harmful enzymes that break down nerves and muscle fibers. With the help of Mfn2, mitochondria carry calpstatin along nerve cells axons to meet muscle cells. There, calpstatin prevents enzymes from destroying delicate synapse connections. But without Mfn2, mitochondria can’t carry the nutrient.
According to Wang, the findings have broad implications. “Mfn2 deficiency or mutations are commonly observed in patients with ALS, peripheral neuropathy, Alzheimer’s disease, and other neurodegenerative diseases in which synaptic loss has long been recognized as a prominent early feature,” he says. “Supplementing Mfn2 may be a common and effective therapeutic approach to treat a wide range of diseases including but not limited muscular disorders, patients with nerve injury and various major neurodegenerative diseases associated with synaptic loss.”




