
by Avery Luedtke, Ph.D., Senior Scientist, MilliporeSigma, 6000 N. Teutonia Ave., Milwaukee, WI 53209
Perovskite Solar Cells
Perovskite solar cells (PSCs) are photovoltaic (PV) devices containing a light-absorbing layer that has the general formula AMX3,[1] and a crystal structure similar to the mineral perovskite (CaTiO3). The advantageous electronic and optical properties of perovskites have contributed to impressive increases in the performance of PSCs since they were first reported with a current maximum power conversion efficiency (PCE) of 25.2%.[2][3] PSCs have become very popular for research, in part because they offer an exciting platform for experimentation, with many options for chemical and engineering tunability. Other attractive properties of PSCs include their ease of fabrication, direct bandgap, high carrier mobility, large absorption coefficient, and long diffusion length.[4]
To learn more about how these semiconductor nanostructures offer new opportunities for tackling clean energy challenges, watch the MilliporeSigma webinar with Professor Parashant V. Kamat.
Webinar: Light Energy Harvesting with Semiconductor Nanostructures
Architecture and Fabrication
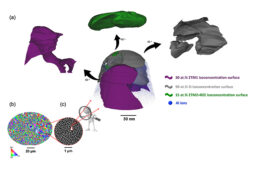
Despite their elegant simplicity, there are various options for the architecture of PSC devices.[5] The transparent electrode can contain the electron transport layer (n-i-p), or the hole transport layer (p-i-n). The layers can be planar with two-dimensional (2D) layer contacts, or contain a mesoporous scaffold. PSCs can also be constructed in tandem with other solar cells, increasing overall efficiency.[6] Simple solution processes, such as spin-coating, are commonly used to manufacture PSC devices, which lend themselves to performance-enhancing modifications, such as solvent and additive engineering.[5][7]
Hole Transport Materials
One of the significant technological advancements in perovskite solar cells was the use of solid hole transport materials (HTM), allowing all solid-state PV devices. One long-used organic HTM, Spiro-MeOTAD (MilliporeSigma Cat. Nos. 792071, 902500), remains very popular for high-performance solar cells; however, many other solution-processable HTMs have been reported for use in PSCs, including organic, metal-organic, and inorganic materials (MilliporeSigma Cat. No. 907642).[8][9][10]
Stability
Although the performance of PSCs is impressive, long-term stability remains a significant research opportunity. Fortunately, recent insights into degradation mechanisms indicate that improvements in device stability can be expected in the coming years.[11]
2D Perovskites
The monovalent cation, A in the formula AMX3, used in the fabrication of a perovskite material is an important choice. Not only does this affect the electronic and optical properties, but also, if a bulky amine is used instead of a three-dimensional (3D) perovskite structure, a layered 2D structure is obtained. These 2D perovskites can be processed in liquid solution or dispersion, and have launched a field of their own. Applications of 2D perovskites include solar cells[12] as well as light-emitting diodes (LEDs)[13], phototransistors [14], and lasers.[15]
Illuminate your research with the latest news and resources on solar cell technology. In our free publication, Material Matters, leading experts address research needs in materials science and nanotechnology. The latest volume, Material Matters 15.2, focuses on recent trends in material discoveries associated with perovskites, and solar cells, as well as battery advances. Dr. Sharma from the Council of Scientific & Industrial Research, and Dr. Correa-Baena from Georgia Institute of Technology, also discuss the latest research and efforts for enhancing performance and stability of perovskite materials.
Material Matters™
Lead-Free Perovskites
The potential impact of lead on the environment and human health complicate its use in commercial solar cell applications, motivating researchers to investigate functional lead-free options for PSCs.[16] Being just above lead in group 14, tin is a discernible lead substitute. The PCE of tin PSCs can be above 7%,[17][18] which is notably lower than that of lead-based PSCs. A different approach that may be of interest to researchers is the use of two different metals, one monovalent and one trivalent, as a modification of the typical perovskite structure.[16] Although synthetically challenging, these double perovskite materials would notably increase the options available for optimization.
Learn more about our latest products in our “Perovskite Solar Cells” brochure.
Perovskite Solar Cells Brochure
1) In this formula, A is a monovalent cation [for example, MeNH3+, HC(NH2)2+, or Cs+], M is a divalent metal (for example, PB2+ or SN2+), and X is a halide (I, Br, or Cl). 2) Kojima, A.; Teshima, K.; Shirai, Y.; Miyasaka, T. Organometal Halide Perovskites as Visible-Light Sensitizers for Photovoltaic Cells J. Am. Chem. Soc. 2009, 131, 6050. DOI: 10.1021/ja809598r 3) https://www.nrel.gov/pv/assets/pdfs/best-research-cell-efficiencies.20200708.pdf (accessed July 23, 2020) 4) Zhang, F.; Zhu, K. Additive Engineering for Efficient and Stable Perovskite Solar Cells Adv. Energy Mater. 2020, 10, 1902579. DOI: 10.1002/aenm.201902579 5) Correa-Baena, J.-P.; Saliba, M.; Buonassisi, T.; Grätzel, M.; Abate, A.; Tress, W.; Hagfeldt, A. Promises and challenges of perovskite solar cells Science 2017, 358, 739. DOI: 10.1126/science.aam6323 6) Walia, Q.; Elumalaic, N. K.; Iqbala, Y.; Uddinc, A.; Joseb R. Tandem perovskite solar cells Renewable and Sustainable Energy Reviews 2018, 84, 89. DOI: 10.1016/j.rser.2018.01.005 7) Mahapatra, A.; Prochowicz, D.; Tavakoli, M. M.; Trivedi, S.; Kumara, P.; Yadav, P. A review of aspects of additive engineering in perovskite solar cells J. Mater. Chem. A, 2020, 8, 27. DOI: 10.1039/c9ta07657c 8) Calil, L.; Kazim, S.; Grätzel, M.; Ahmad, S. Hole-Transport Materials for Perovskite Solar Cells Angew. Chem. Int. Ed. 2016, 55, 14522. DOI: 10.1002/anie.201601757 9) Urieta-Mora, J.; García-Benito, I.; Molina-Ontoria, A.; Martín, N. Hole transporting materials for perovskite solar cells: a chemical approach Chem. Soc. Rev., 2018, 47, 8541. DOI: 10.1039/c8cs00262b 10) Singh, R.; Singh, P. K.; Bhattacharyac, B.; Rheea, H.-W. Review of current progress in inorganic hole-transport materials for perovskite solar cells Applied Materials Today 2019, 14, 175. DOI: 10.1016/j.apmt.2018.12.011 11) Wali, Q.; Iftikhar, F. J.; Khan, M. E.; Ullah, A.; Iqbal, Y.; Jose, R. Advances in stability of perovskite solar cells Organic Electronics 2020, 78, 105590. DOI: 10.1016/j.orgel.2019.105590 12) Cao, D. H.; Stoumpos, C. C.; Farha, O. K.; Hupp, J. T.; Kanatzidis, M. G. 2D Homologous Perovskites as Light-Absorbing Materials for Solar Cell Applications J. Am. Chem. Soc. 2015, 137, 7843. DOI: 10.1021/jacs.5b03796 13) Yuan, M.; Quan, L. N.; Comin, R.; Walters, G.; Sabatini, R.; Voznyy, O.; Hoogland, S.; Zhao, Y.; Beauregard,E. M.; Kanjanaboos, P.; Lu, Z.; Kim, D. H.; Sargent, E. H. Perovskite Energy Funnels for Efficient Light-Emitting Diodes" Nat. Nanotechnol. 2016, 11, 872. DOI: 10.1038/NNANO.2016.110 14) Shao, Y.; Liu, Y.; Chen, X.; Chen, C.; Sarpkaya, I.; Chen, Z.; Fang, Y.; Kong, J.; Watanabe, K.; Taniguchi, T.; Taylor, A.; Huang, J.; Xia, F. Stable Graphene-Two-Dimensional Multiphase Perovskite Heterostructure Phototransistors with High Gain Nano Lett. 2017, 17, 7330. DOI: 10.1021/acs.nanolett.7b02980 15) Raghavan, C. M.; Chen,T.-P.; Li, S.-S.; Chen, W.-L.; Lo, C.-Y.; Liao, Y.-M.; Haider, G.; Lin, C.-C.; Chen, C.-C.; Sankar, R.; Chang, Y.-M.; Chou, F.-C.; Chen, C.-W. Low-Threshold Lasing from 2D Homologous Organic−Inorganic Hybrid Ruddlesden−Popper Perovskite Single Crystals Nano Lett. 2018, 18, 3221. DOI: 10.1021/acs.nanolett.8b00990 16) Giustino, F.; Snaith, H. J. Toward Lead-Free Perovskite Solar Cells ACS Energy Lett. 2016, 1, 1233. DOI: 10.1021/acsenergylett.6b00499 17) Liu, J.; Ozaki, M.; Yakumaru, S.; Handa, T.; Nishikubo, R.; Kanemitsu, Y.; Saeki, A.; Murata, Y.; Murdey, R.; Wakamiya, A. Lead-Free Solar Cells based on Tin Halide Perovskite Films with High Coverage and Improved Aggregation Angew. Chem. Int. Ed. 2018, 57, 13221. DOI: 10.1002/anie.201808385 18) Li, F.; Zhang, C.; Huang, J.-H.; Fan, H.; Wang, H.; Wang, P.; Zhan, C.; Liu, C.-M.; Li, X.; Yang, L.-M.; Song, Y.; Jiang, K.-J. A Cation-Exchange Approach for the Fabrication of Efficient Methylammonium Tin Iodide Perovskite Solar Cells Angew. Chem. Int. Ed. 2019, 58, 6688. DOI: 10.1002/anie.201902418
Sponsored content by MilliporeSigma






Good one.
Webinar on such topic of Research and development are highly useful