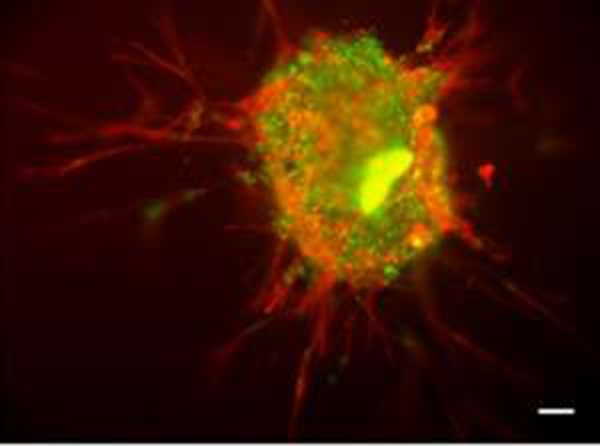
Activated stem cells exiting and migrating out of the spheroid towards an injury. (Credit: University of Glasgow)
Scientists have discovered a new way to replicate the regenerative power of stem cells in the lab, which could lead to powerful treatments for injuries and diseases.
In a new paper published in the journal ACS Nano, Catherine Berry, Ph.D., and Prof. Matthew Dalby from the University of Glasgow‘s Institute of Molecular Cell and Systems Biology, describe how they have been able create and control bundles of bone marrow stem cells that act as the stem cells do in the body, These bundles of cells are capable of healing injuries in lab-grown bone and cartilage models.
Mesenchymal stem cells are produced naturally in the body and have the unique ability to develop, or ‘differentiate’, into many other different types of cells, such as bone, cartilage or fat cells. They have enormous potential for use in medicine but they are difficult to properly culture in the laboratory, because when stored outside of the body they spontaneously and randomly differentiate.
In the body, however, they wait in the bone marrow until a regenerative demand is placed on them. Then, they change into mature cell types to repair the bone, cartilage, ligament or tendon around them.
By using 3D spheroid cultures rather than standard 2D cell growth techniques, the researchers have shown that they can grow stem cells and prevent them from differentiating over a prolonged period.
To achieve this, they add magnetic nanoparticles to each stem cell. Then, using a simple magnet, they built ball-shaped masses of around 10,000 stem cells, just big enough to see with the naked eye, over the course of just a few hours. When placed in soft gel, similar to the consistency of bone marrow jelly, these balls of cells remain as stem cells over long culture periods, with no unwanted differentiation
Next, the researchers placed the 3D spheroid cultures next to laboratory wound models of cartilage, bone and ligament. When the bundle of stem cells were placed next to the injured area, they quickly became active again, migrated towards the injured tissue, and began to differentiate in the same way they would in the human body to heal a wound.
As well as for understanding regeneration of the musculoskeletal system, the tiny marrow stem cell models could be used to test drugs designed to activate the stem cells and have applications beyond bone, cartilage and ligament repair.
Dr Catherine Berry, of the University’s Institute of Molecular Cell and Systems Biology, said: “This is a really exciting discovery, which uses a fairly simple and affordable method to grow and maintain stem cells ready to heal tissues.
“Although this paper is on our research into using these stem cells with bone and cartilage, we’re already starting to work with partners at the Paul O’Gorman Leukaemia Research Centre and the Beatson West of Scotland Cancer Centre to look at the potential to use this technique to combat leukaemia and breast cancer.
“Both diseases can hide bundles of cancer cells in the bone marrow which lay dormant for years before being becoming active again, causing secondary cancers which are often very difficult to treat, It may be that changes in mesenchymal stem cell activity are linked to cancer cell activation.
“We’re keen to explore how we can use our technique to understand more about how stem cells communicate with other cells and what we can do to use stem cells more effectively in medicine.”
The paper, titled ‘A Quiescent, Regeneration-Responsive Tissue Engineered Mesenchymal Stem Cell Bone Marrow Niche Model via Magnetic Levitation’, is published in the journal ACS Nano.




