Researchers from the University of Michigan have discovered an important new facet of programmed cell death. Recent studies have revealed that proteins involved in the process assemble into channels that shuttle calcium ions. By itself, this isn’t enough to initiate cell death. The team has now discovered the vital next step. The team worked with Arabidopsis and Nicotiana benthamiana, popular plant model systems, and a high-resolution total internal reflection fluorescence microscope to make the discovery, which they published in Nature.
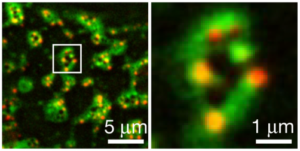
Images from a high resolution microscope at the University of Michigan help reveal a never-before-observed ring structure that researchers have found helps cells mediate the “perfect” amount of cell death. The green parts of the ring show where it is bound to a cell membrane and the red dots are channels that penetrate through the membrane. Credit:D. Ge et al. Nature, 2026. DOI:10.1038/s41586-026-10215-1
The channels are organized into a ring structure on the cell membrane. The ring is a combination of proteins that bind to the cell membrane and six channels that orient themselves to traverse the membrane. The purpose of the ring structure is still unknown. The team hypothesizes that the rings enable communication with nearby cells, sending inflammation signals that initiate targeted cell death. The researchers are working with the U-M Life Sciences Institute and its Cryo-Electron (Cryo-EM) Lab to examine the rings in greater detail.
“The next thing that we’re doing is looking at what kinds of things could be leaking out through this structure, and also what supports the ring structure formation,” Shan said. “We haven’t answered all the questions, but we have advanced the field.”
Integrating TIRF and cryo-EM workflows to resolve the six-channel geometry
The researchers used total internal reflection fluorescence (TIRF) microscopy to observe the in vivo formation of the rings at the cell periphery in real time. Standard confocal microscopy often lacks the signal-to-noise ratio to distinguish between membrane-bound protein clusters and the noise of the cytoplasm. In contrast, TIRF only excites fluorophores within 100 nanometers of the plasma membrane. This allowed the researchers to observe the live assembly of the proteins.
This marked the first time the scientists could see the proteins not just as calcium channels, but as organized punctate structures forming specifically at the membrane interface during a pathogen attack.
While TIRF showed where and when the rings form, Cryo-EM reveals how they are built. By flash-freezing the protein complexes, the team could capture the proteins in their active resistosome state. The Cryo-EM workflow allowed the researchers to resolve the six-channel geometry. Instead of a single protein pore, they discovered a higher-order assembly of six individual channels organized into a single circular unit.
The discovery could open up new avenues for scientists working to make plants more resilient and to treat conditions that involve cell death in humans.
“We know there are a lot of unknowns with this ring hanging on the ceiling of cells, but we know it is absolutely required to have the perfect amount of cell death, to have the perfect immune response,” Shan said. “We truly believe this work will lay the foundation to launch a wave of exciting research for continued discovery.”