[Adobe Stock]
Zoom out, and the clinic, in a sense, is in its own waiting room. For instance, site activation cannot start until the contract is signed. Meanwhile, the contract cannot finalize until the budget settles, and the budget cannot settle until the review board approves.
Take a step back and drug development is a stack of waiting rooms, too, each one bigger than the one inside it. A patient waits for a test. A site waits for activation. A protocol waits for evidence that its criteria can enroll the patients it specified. A molecule waits for each phase to produce enough proof for the next. A drug, even after approval, waits for coverage.
It’s common industry knowledge that the total clock from discovery takes ten years or longer with the majority of candidates never making it to the end. For those that do, the clock is really an accumulation of smaller pauses: evidence gaps, enrollment delays, site bottlenecks, protocol misfires and access questions that surface too late. The FDA’s basic map of drug development runs from discovery and preclinical testing through clinical research, FDA review and postmarket monitoring. Each step asks for a different kind of proof.

Suzanne Caruso
A growing roster of AI vendors says it can loosen that logjam. Norstella’s bet is that the fastest way through the maze is to pressure-test trial decisions before the maze closes around them. Its vehicle is NorstellaLinQ, a linked data asset built to surface those decisions earlier.
AI use in pharma is well past the pilot stage. A 2024 Norstella survey found that 81% of senior life sciences decision-makers said their organization is already using AI in at least one development program. The maturity is gradually building. “We’re starting to see little wins there, but we are in the infancy of seeing those wins, and they’re very tactical,” said Suzanne Caruso, Norstella’s general manager and executive vice president of clinical, regulatory and strategic intelligence. Caruso says Norstella sees more “workflow wins” and that sponsors are “taking administrative tasks that have a lot of data or a lot of handoffs, and consolidating them.” The result is getting to the next decision faster.

The bet before the bet
Historically, one of the traditional clinical trial bottleneck drivers was the difficulty of gaining a horizontal view across trials. Consider that a significant number of clinical trial principal investigators run few trials in their careers. A 2017 study found that just over half ran a single one.

Kris Kaneta
But if you analyze trials at large, clear bottlenecks emerge. “If you really zoom out and think about where the largest potential is going to be, my position is, it’s going to be before the decision even gets made, before the trial even starts, before a molecule even receives that two billion dollar commitment,” said Kris Kaneta, Norstella’s chief product officer.
The biopharma sector is unique in the share of resources it invests in new therapies when grappling simultaneously with a significant failure rate. In Kaneta’s framing, placing “higher-confidence bets” starts before the first patient is enrolled.
Unifying knowledge threads
The assumptions that guide a trial must be analyzed in context. A clinical team may know which endpoints have worked before. A feasibility team may know where patients are treated. A commercial team may know whether the market is crowded. A payer team may know whether access will be difficult. The challenge is that each answer usually sits in a different system, with a different vocabulary and a different owner.
It doesn’t help that clinical development has clung to spreadsheets for years. “I cannot tell you the amount of clinical work that is still done in Excel,” Caruso said. The transition then is a move from spreadsheets alone to linked data sets and recommendation engines. “It’s no longer just a flat, point-in-time dataset. AI can look across a dataset, see the trends underpinning it, and say, based on this and everything done prior, let me make recommendations on the next best action,” she said.
A linked data foundation
Norstella’s approach to unifying these threads is NorstellaLinQ, launched in October 2024 and now central to how the company frames its AI strategy. The asset links real-world data, including claims, labs and structured and unstructured EMRs, with Norstella’s proprietary forecasting, clinical, regulatory, payer, coverage and commercial intelligence. Norstella says LinQ spans more than 74 billion data points and draws from Citeline, Evaluate, MMIT, Panalgo and The Dedham Group.
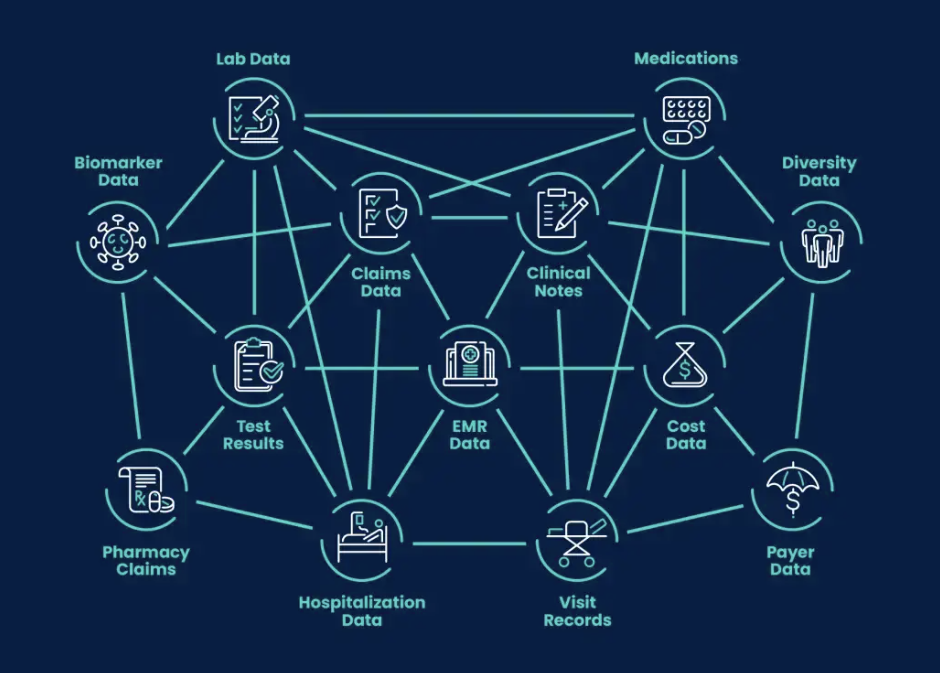
A marketing image from Norstella showing how its NorstellaLinQ data asset integrates real-world data and its proprietary intelligence. [Image from Norstella]
That work demands considerable plumbing. It requires ontologies, knowledge graphs, entity extraction and people who know the difference between terms that look distinct to a machine but mean the same thing in clinical practice. Kaneta described the role of subject-matter experts as indispensable because they understand “what one term means and how it’s actually the same thing as another term, but it just happens to be documented a different way.”
Circling back to trust
If AI is only summarizing a document, a weak answer wastes time. But if AI is shaping protocol design, site selection or investigator recommendations, a weak answer can distort the trial itself. “Truthfully, it doesn’t start out perfect,” Caruso said. “It takes a lot of manual work of humans training the model.”
Kaneta described the evaluation problem as moving from a binary question to a more difficult one. AI models are evolving and growing more sophisticated. As a result, a model can produce an answer that is plausible, well sourced and still incomplete for the trial at hand. “We are entering into degrees of correctness,” he said.
That is why Norstella keeps coming back to context. “I am trying to understand and leverage the best possible models in the context of our 74 billion real-world data points and our two decades of historical clinical trial data,” Kaneta said.
Caruso said the other half of the trust equation is provenance. “I can’t just give a report without any citations anymore,” she said.


Tell Us What You Think!
You must be logged in to post a comment.