Scientists at the Department of Energy’s (DOE) Argonne National Laboratory have revealed the molecular movement of lanthanide separation, an essential step in rare earth element extraction. Their study was published in Chemical Science.

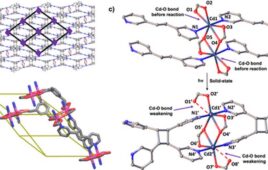
Illustration capturing the “molecular dance” of lanthanide separation. Credit: Argonne National Laboratory
The team studied lanthanides, a rare earth element with a wide variety of uses, from TV screens to oil refining. The DOE classifies lanthanides as critical materials because of their importance and the supply risk, explained Michael Servis, an Argonne chemist.
Lanthanides are a group of 15 metallic elements, atomic numbers 57-71. They are essential for technologies like magnets, but they are often found in ores, which makes them difficult to separate. Lanthanides are typically separated through solvent extraction, where they are dissolved in an acidic solution and selectively separated into an oil phase.
Metadynamic simulation
The team used advanced computer simulations to reveal the choreography of molecules during the lananthide extraction process. They found that extractant molecules – other ions and water molecules – surround the lanthanide ions and try to bond with them. The molecule that binds with the ion determines which element gets separated during extraction.
Using a simulation technique called metadynamics, the researchers created a map of the energy landscape during the extraction process. “Metadynamics helps us see all the possible ways molecules can arrange themselves around the lanthanide. This technique gives us clues about why some lanthanides are easier to separate than others,” said Servis.
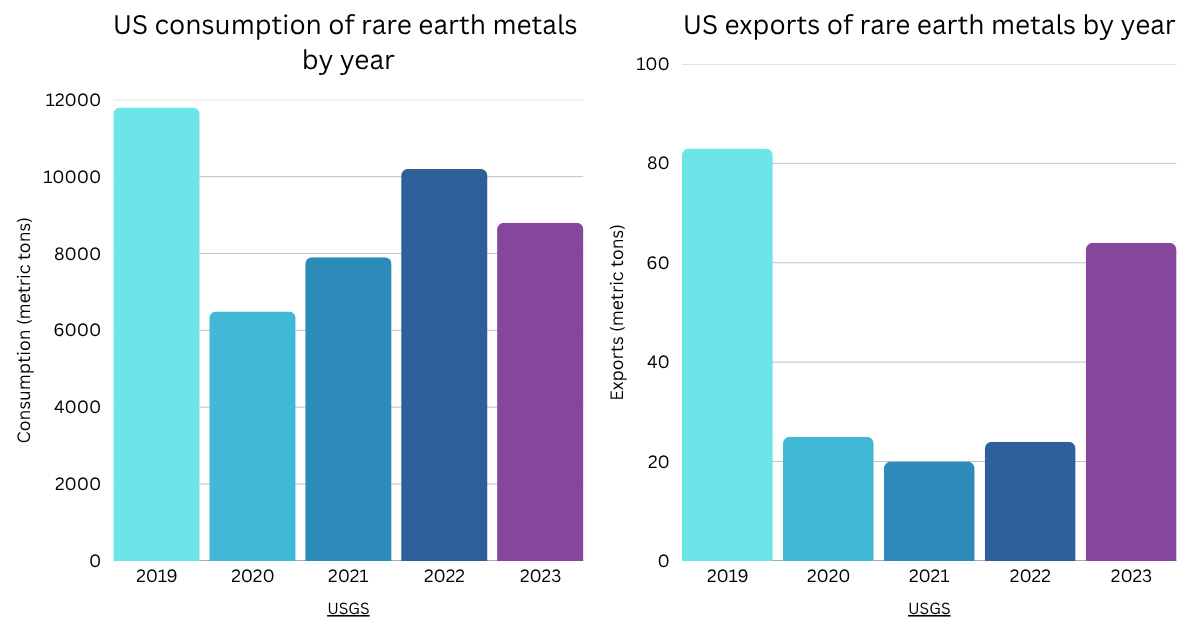
US consumption and exports of rare earth metals. Data from USGS
The scientists found that lighter lanthanides, such as lanthanum and europium, form stronger bonds with the extractant molecules than heavier lanthanides, which struggle due to crowding.
Revealing other factors
The water molecules also play an important role in this process. Some water molecules bind to the lanthanide ion and help stabilize interactions by forming hydrogen bonds. This expands the possible movements of the molecules.
The simulations showed how ligand crowding at the metal center can control selectivity as it causes Lu – the lightest lanthanide – to lose coordination with the extractant, resulting in an extraction trend opposite to the current system. The simulation separates lighter molecules more easily than heavy molecules, which is the opposite of the current system.
This study could pave the way for more efficient extraction and separation of rare earth elements. The team is exploring other solvents and extractant molecules to improve the selectivity of their process.