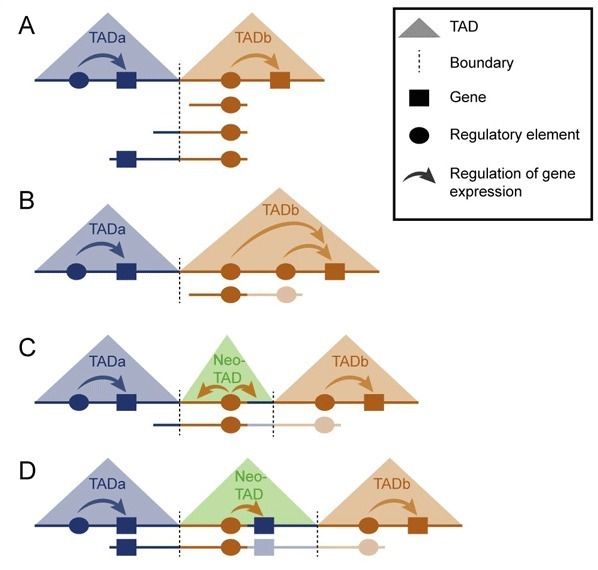
A) Diagram of two TADs in a healthy genome; B) The duplication occurs entirely within a TAD (intra-TAD), and the higher-level structure of the genome is unaffected. Misregulation of the duplicated gene occurs within the TAD; C) The duplication includes the boundary between two TADs (inter-TAD), resulting in a new TAD (neo-TAD) that contains only regulatory elements but no gene, so that no effects are exerted on the organism; D) The duplication includes the boundary between two TADs (inter-TAD) and a gene in the neighboring TAD. The resulting neo-TAD contains the regulatory elements/information of the orange-colored TAD and a gene from the blue TAD, which is consequently misregulated. Image: © MPI for Molecular Genetics
Duplications of DNA segments are common in the human genome. Duplications have been proposed as an evolutionary mechanism but they can also be associated with human disease. Scientists from the Max Planck Institute for Molecular Genetics and the Charité-Universitätsmedizin Berlin have now shown that, depending on their position in the genome, duplications can lead to the formation of new functional units known as topologically associated domains (TADs). In the journal Nature, the researchers describe how the duplication of DNA segments of various sizes gives rise to new TADs and how duplicated genes can acquire new functions. This mechanism represents a possible mechanism how new gene functions can arise in the course of evolution and has far-reaching implications for the interpretation of genetic mutations in humans.
Topologically associated domains — or TADs for short — are spatially separated regions in the genome. They are DNA segments that are a million base pairs long on average and contain one or more genes and their regulatory elements, called enhancers. The latter control when and in which cells a gene is switched on or off. TADs are separated from each other by boundary elements, so that gene activity in one TAD is independent from that in neighboring TADs.
By studying duplications of various sizes in the region of the SOX9 gene, scientists of the Research Group Development & Disease at the Max Planck Institute for Molecular Genetics have discovered a previously unknown function of TADs. The SOX9 gene is responsible for the expression of a transcription factor that has important functions in skeletal and male sexual development. By analyzing the three-dimensional structure of the genome, the researchers were able to show that two large TADs are located in the SOX9 region. One of them contains the SOX9 gene and its regulatory elements; the other contains two genes, KCNJ2 and KCNJ16, two potassium channels, and their regulatory elements.
Duplications in the region of the SOX9 gene can lead to radically different clinical pictures. In order to understand these differences, the researchers investigated three duplications, all of which include regulatory elements of the SOX9 gene as well as other DNA segments of various lengths.
The first of the duplications examined leads in humans to a reversal of gender from female to male. Affected individuals are morphologically male despite possessing two X chromosomes. The scientists showed that this duplication only affects the DNA and regulatory elements that occur within the SOX9 TAD. Surprisingly, however, larger duplications that include this region but extend up to the neighboring genes KCNJ2 and KCNJ16 and therefore contain sections of the adjacent TAD have no clinical effects on sex determination. By contrast, even bigger duplications that include not only the non-coding DNA in the SOX9 region but also the neighboring KCNJ2 gene lead to Cooks syndrome, a hereditary abnormality of the hands and feet characterized by the absence of fingernails and shortened fingers and toes.
“We have to distinguish between intra-TAD and inter-TAD duplications, depending on their location,” says Stefan Mundlos, head of the research group that carried out the work. “In the case of sex reversal, regulatory elements within a TAD (intra-TAD) are duplicated, and the effect remains limited to that TAD. Thus, only the gene in the TAD is misregulated, meaning that it is activated either too much or too little. In the case of the SOX9 gene, the activity of the gene is increased, resulting in the development of the male gender, even if the affected individual is genetically female.”
Because TADs are functionally separated from each other, intra-TAD duplications only affect one TAD and have no impact on neighboring TADs. In the case of inter-TAD duplications, by contrast, not only the regulatory elements of the SOX9 region are duplicated, but also the boundary between two TADs. The duplicated boundary defines a new TAD (called neo-TAD) that includes the duplicated region of the genome and insulates it from the rest of the genome.
“The effects of a neo-TAD depend on which elements of the genome it contains,” says Mundlos. “If the neo-TAD contains only regulatory elements but no genes, it has no effects on the organism. Neighboring genes are shielded from the regulatory elements by the TAD boundaries and remain unaffected. However, if a neo-TAD contains both regulatory elements and a gene from a neighboring TAD, the latter can be regulated by the regulatory elements (enhancer) within the neo-TAD, resulting in gene misexpression, which might trigger abnormalities or disease.
This was exactly the case for the third duplication studied by the researchers, which triggers Cooks syndrome. The new TAD contains not only the regulatory elements of the SOX9 gene, but also a copy of the KCNJ2 gene. The KCNJ2 gene in the neo-TAD is then regulated by the regulatory elements of the SOX9 gene. As a result, it is activated at the wrong time and in the wrong tissues during development, giving rise to the abnormalities associated with this syndrome.
By including the TAD structure in the interpretation of genetic alterations, scientists can make much more precise predictions about the effects of mutations than it was previously the case. This relates, for example, to duplications that cause hereditary diseases as well as alterations in cancer cells, in which duplications commonly occur.
Duplications are one of the driving forces of evolution. The duplication of a gene makes it possible for the copy to develop new and different properties from the original gene, which remains unaffected by this process. By enabling new combinations of regulatory elements and genes in neo-TADs, duplications could represent an evolutionary mechanism for the emergence of new gene functions.



