Scientists at Johns Hopkins Medicine have developed a simplified version of biodegradable nanoparticles that can teach the immune system to find and destroy disease-causing cells in the body. They published their findings in Science Advances.
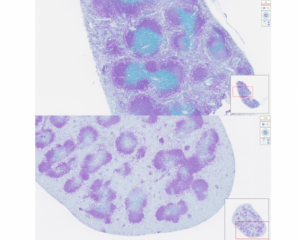
The Johns Hopkins-developed nanoparticles were able to induce the destruction of the majority of the immune system cells called B cells, which contribute to diseases such as lupus and cancers of the blood. Top image: Blue dyed B cells in the spleen of an untreated mouse. Bottom image: Reduction of blue B cells in the spleen of a mouse treated with the nanoparticles. Credit: Manav Jain and Jordan Green, Johns Hopkins Medicine
Engineered immune cells have been successfully used to treat an array of blood cancers, using CAR-T cells or chimeric antigen receptor T cells. The treatment works by drawing immune T cells from a patient’s blood and engineering the cells in the laboratory to have receptors that recognize and kill cancer cells. However, the process to remove the blood cells and individually engineer them outside of the body is costly and inefficient.
The new study describes nanoparticles that were engineered to travel to and stimulate immune T cells to seek out and destroy other immune system cells called B cells, which are the source of diseases such as lupus, leukemia and lymphoma. The nanoparticles contain mRNA that instructs T cells to express receptors on their surface that detect cancer and lupus-causing B cells.
The nanoparticles are composed of polymers with ester units that biodegrade in water. The nanoparticle surface features antiCD3 and antiCD28, antibody molecules that help the nanoparticles find and stimulate T cells.
The particles act as a staged delivery vehicle. First, they target and activate the T cells. Once the T cells are activated, they internalize the particle. After this, the polymer backbone biodegrades, releasing the mRNA to reprogram the cell.
A streamlined 3-component polymer architecture replaces complex lipid systems
Other researchers have developed lipid-based nanoparticles with five components. Comparatively, the Johns Hopkins nanoparticles feature a simpler design requiring only three components. Additionally, the nanoparticles are pH-responsive. As such, they are stable at physiological pH but degrade rapidly once they reach the acidic environment of the cell’s endosome, resulting in a 10% cargo delivery rate.
The JHM study shows that 95% of the target B cells were depleted in the circulating blood of healthy mice 24 hours after injection. About 50% of B cells were destroyed in the spleens of the mice. After a week, B cells in the blood returned to about 50% of their original quantity.
“These experiments were successful using just one dose of the nanoparticles, and an advantage of using an off-the-shelf therapy is the potential for scalable manufacture and broad accessibility, whereas current forms of CAR-T therapies are very expensive and time-consuming,” says Jordan Green, Ph.D., the Herschel L. Seder professor of biomedical engineering at the Johns Hopkins University School of Medicine.
Ph-responsive behavior and endosomal escape drive 10% delivery efficiency
The researchers found that the degradable nanoparticles worked just as well as commercially made magnetic beads designed to latch on to T cells for laboratory research purposes. The nanoparticles were also able to enter the T cells to reengineer them.
In a previous study, Green and his colleagues found that about 10% of the Johns Hopkins-developed nanoparticles successfully escape the cell’s degradation compartments to deliver their sensitive genetic cargo, compared with 1% to 2% of other nanoparticles that immediately get degraded and ejected from the cell.
The researchers say the Johns Hopkins research team aims to continue refining the nanoparticles, tailoring them better to diseased B cells and able to dial up or down the amount of T cell stimulation.