 C. Light Technologies, a neurotech and AI company, announces it has received an investment from the Alzheimer’s Drug Discovery Foundation (ADDF) through its Diagnostics Accelerator, a collaborative research initiative supported by Leonard A. Lauder, Bill Gates, the Dolby family, the Charles and Helen Schwab Foundation, Jeff Bezos and MacKenzie Scott among other leading philanthropists. This new investment will fund a novel pilot study with the UCSF Memory and Aging Center to look at changes in fixational eye motion during the earliest stage of Alzheimer’s disease, generally known as mild cognitive impairment.
C. Light Technologies, a neurotech and AI company, announces it has received an investment from the Alzheimer’s Drug Discovery Foundation (ADDF) through its Diagnostics Accelerator, a collaborative research initiative supported by Leonard A. Lauder, Bill Gates, the Dolby family, the Charles and Helen Schwab Foundation, Jeff Bezos and MacKenzie Scott among other leading philanthropists. This new investment will fund a novel pilot study with the UCSF Memory and Aging Center to look at changes in fixational eye motion during the earliest stage of Alzheimer’s disease, generally known as mild cognitive impairment.
The ADDF’s Diagnostics Accelerator has made previous investments in more than two dozen world-class research programs to explore blood, ocular and genetic biomarkers, as well as technology-based biomarkers to identify the early, subtle changes that happen in people with Alzheimer’s. As new therapeutics for Alzheimer’s are introduced to the clinic, this UCSF technology has the potential to provide clinicians a better method to measure disease progression, and ultimately therapeutic efficacy, using C. Light’s novel retinal motion technology.
The lack of objective tools to assess neurological progression and disability highly impacts pharmaceutical developers. Therapeutic development for the central nervous system is extremely difficult, with success rates of only 8% to go from conception to market. Clinical trials can take a decade to come to fruition in this space due to inefficient methodologies to assess the efficacy of drugs. This is where C. Light could make the biggest impact.
“Before this year, it had been almost 20 years since an Alzheimer’s drug was brought to market, and part of the reason for this very slow progress is that drug developers haven’t had viable biomarkers that they can use to effectively stratify patients and track disease on a fine scale. The ADDF’s investment will allow us to do that,” said Dr. Christy K. Sheehy, co-founder of C. Light.
Eye motion has been used for decades to quickly triage brain health, which is why a doctor asks you to “follow my finger” with your eyes. In more than 30 years of research, studies have revealed that Alzheimer’s disease patients’ eye movements are affected by the disease, though to date, these eye movements have only been measured on a larger scale.
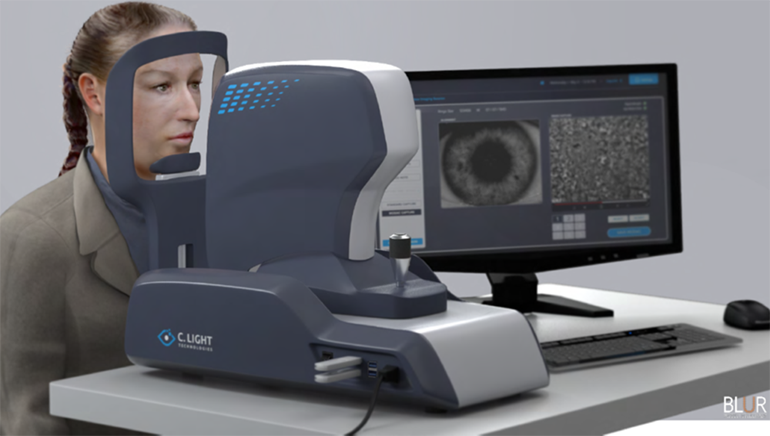
C. Light’s research takes the eye movement tests to the microscopic level for quicker and earlier assessments. C. Light’s fast, non-invasive, and objective technology enables clinicians to study and measure eye motion on the cellular scale – meaning eye motion as small as 1/100th the size of a human hair – helping physicians to better monitor a patient’s disease and treat it more effectively.
C. Light’s tests are easy for clinicians to administer. Patients put their chin in a chinrest and then focus on a target for 10 seconds. The test does not require eye dilation, and patients are permitted to blink during the test. A very low-level laser light is shown through the pupil and reflects off the patient’s retina, while a sensitive camera records the cellular-level motion in a high-resolution video. This eye motion is then extracted up to 1000 times per second and fed into C. Light’s advanced analytical platform.
“C. Light is creating an entirely new data stream about the status of brain health via the eye,” Sheehy continued. “Our growing databases and accompanying AI can change the way we monitor and treat neurological disease for future generations. Ultimately, we’re working to increase the longevity and quality of life for our loved ones.”
C. Light recently completed its second and final seed round raising $500,000, including the ADDF investment, which brings their total seed funding to more than $3 million. Second round seed funders included: ADDF, the Wisconsin River Business Angels, Abraham Investments, LLC and others.
“Over 6 million people in the U.S. are affected by Alzheimer’s. Technology is now capable of collecting data from patients in many different ways, including using AI to find patterns that detect early signs of dementia,” said Howard Fillit, M.D., founding executive director and chief science officer at the ADDF. “C. Light’s retinal measurement solution along with the UCSF study will help clinicians identify deficits and may allow for early intervention and quicker therapeutic intervention. This has the potential to help find more precise treatments for patients.”