
Altaviva image courtesy of Medtronic
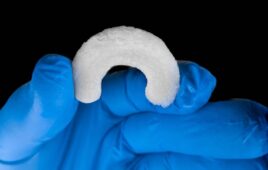
The medical device giant Medtronic won approval for its Altaviva implant, a tibial neurostimulator, in September. The device, roughly half the length of a stick of chewing gum (43.7 × 15.7 × 4.5 mm, 7.5 g), recently won a 2026 Edison Award for health innovation. Medtronic enrolled its first patient in the ENDURANCE post-approval study. “As Altaviva is a Class 3 device, we’re required to follow those patients for a period of time,” said Emily Elswick, president of Medtronic Pelvic Health. “We’ll be following them for five years. The primary endpoint is the proportion of patients experiencing a 50% or greater improvement in UUI (urge urinary incontinence) episodes.”
The company is betting the device can reach a population that existing therapies have largely missed. An estimated 43 million U.S. adults have bladder control problems. Of those, roughly 16 million have UUI, a common symptom of overactive bladder characterized by sudden, intense urgency followed by involuntary leaking. “Sadly, the market for adult diapers is growing faster than the market for baby diapers,” Elswick said.
An implantable alternative to weekly office visits
 The Altaviva device is implanted near the ankle, above the fascia and anterior to the Achilles tendon, in a procedure requiring no sedation and no imaging. Medtronic projects that the device has a 15-year battery life under standard settings. The standard “default” setting is typically 30 minutes per day, 3 days per week. It requires recharging only once or twice a year. Patients with an Altaviva device can receive MRI scans anywhere on the body per device labeling.
The Altaviva device is implanted near the ankle, above the fascia and anterior to the Achilles tendon, in a procedure requiring no sedation and no imaging. Medtronic projects that the device has a 15-year battery life under standard settings. The standard “default” setting is typically 30 minutes per day, 3 days per week. It requires recharging only once or twice a year. Patients with an Altaviva device can receive MRI scans anywhere on the body per device labeling.
The method of therapy itself is well established. Stimulating the tibial nerve to treat bladder symptoms has been a recommended third-line treatment in AUA/SUFU guidelines for over a decade through percutaneous nerve stimulation, essentially acupuncture-style needles requiring weekly office visits for 12 weeks and ongoing maintenance after that. Medtronic set out to make that therapy implantable. The development took about five years from concept through the TITAN 2 pivotal trial (NCT05226286), which enrolled 126 implanted patients across 26 U.S. sites. “Now that we have the TITAN 2 data published, our healthcare economics, policy, and reimbursement team are out talking to carriers about commercial coverage,” Elswick explained. Medtronic implanted its first commercial patient within four days of approval.
-

Emily Elswick
“Being able to give a patient relief the day they receive the implant is unique to us, and really important for them,” Elswick said. “These patients have probably gone many years either suffering in silence or wearing diapers or pads, dealing with medications that may or may not have worked or that weren’t financially possible for them.”
Tracking 170 patients over five years
ENDURANCE, formally titled the Post Approval Effectiveness and Durability Evaluation of the Altaviva Tibial System, will follow 170 implanted patients at up to 30 U.S. centers over five years. The first procedure was performed by Vincent R. Lucente, MD at Axia Women’s Health in Allentown, Pennsylvania. At least 50% of enrolled patients must be aged 65 or older, a requirement that traces directly to what the FDA found in the pivotal data.
In the TITAN 2 pivotal study, the study population was predominantly female (95%), with a mean age of 62.6 years. All 126 patients had failed at least two overactive bladder medications before enrollment. Roughly 18% had previously tried Botox injections, in which onabotulinumtoxinA is injected directly into the bladder wall to calm overactive muscle contractions that trigger urgency and leaking. Another 9% had tried percutaneous tibial nerve stimulation, according to the FDA’s Summary of Safety and Effectiveness Data.
 At six months, 58.3% of implanted patients achieved at least a 50% reduction in daily UUI episodes, the study’s primary endpoint. That cleared the FDA’s recommended 50% success benchmark. On average, patients went from 5.1 UUI episodes per day at baseline to 2.5 at 12 months, and about 22% achieved complete resolution of urgent leaks. All 30 device- or procedure-related adverse events through 12 months were graded mild or moderate. No device migration or erosion was reported.
At six months, 58.3% of implanted patients achieved at least a 50% reduction in daily UUI episodes, the study’s primary endpoint. That cleared the FDA’s recommended 50% success benchmark. On average, patients went from 5.1 UUI episodes per day at baseline to 2.5 at 12 months, and about 22% achieved complete resolution of urgent leaks. All 30 device- or procedure-related adverse events through 12 months were graded mild or moderate. No device migration or erosion was reported.
Medtronic intentionally designed the Altaviva system to be as unobtrusive as possible for long-term use. “The team was really focused on reducing the cognitive burden for clinicians and patients,” Elswick said. She noted that developers incorporated automatic intelligence into the device early on, such as an internal clock that adjusts on its own for daylight saving time. Medtronic factored in “little things, because you want the therapy to work in the background for 15 years,” Elswick explained. “Patients should know they’re getting relief without constantly being reminded they have an implant.”
While Medtronic has “been in neuromodulation for incontinence for 30 years” with its InterStim product line, Elswick noted that the industry is still only “reaching a very small percentage of the population.” She sees two forces converging to change that. The first is a generational shift in which patients are more willing to seek help rather than manage symptoms in silence. Second, there is a broader pivot in medtech investment away from cardiovascular and robotics toward the nervous system. “The next 10 years may really be about the decade of the brain,” Elswick said. The focus could include “how the brain, the bladder, and the nervous system signals can be regulated to improve conditions,” she concluded.

Tell Us What You Think!
You must be logged in to post a comment.