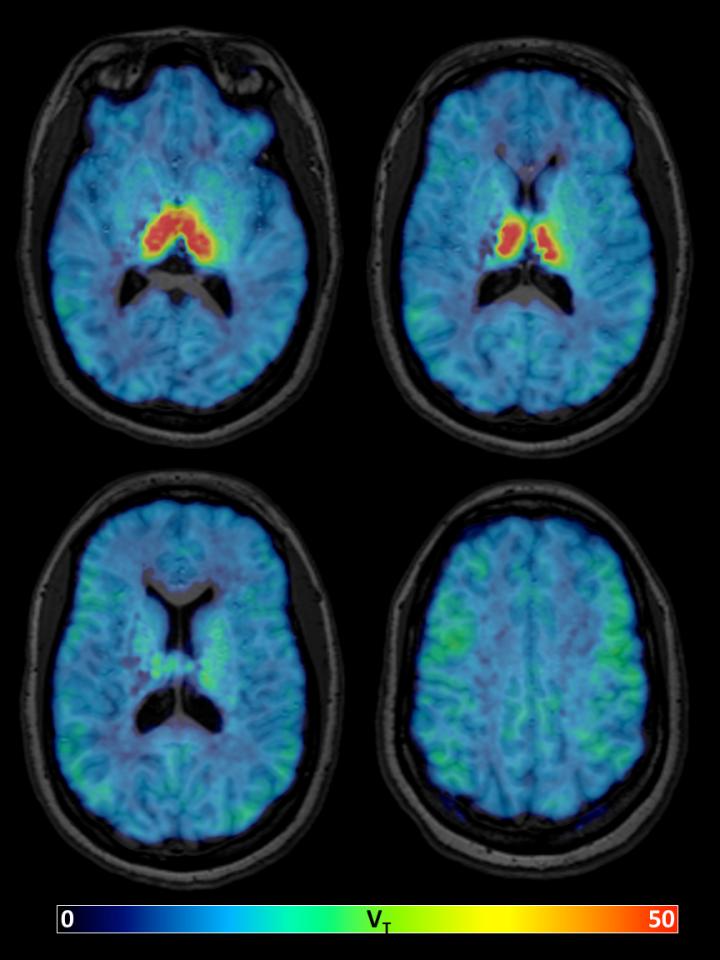
Parametric images of the total distribution volume (VT) of 18F-XTRA, estimated using Logan graphical analysis with metabolite-corrected arterial input function and 90-minute data from one representative healthy participant.
A new PET imaging radiotracer could help researchers understand neurodegenerative disease and the aging brain. The study is featured in the October issue of The Journal of Nuclear Medicine.
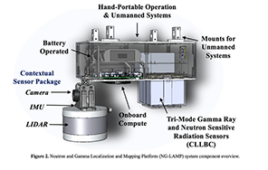
Past studies have shown a reduced density of the α4β2 nicotinic acetylcholine receptor (α4β2-nAChR) in the cortex and hippocampus of the brain in aging patients and those with neurodegenerative disease. The α4β2-nAChR is partly responsible for learning, and even a small loss of activity in this receptor can have wide-ranging effects on neurotransmission across neural circuits. However, fast and high-performing α4β2-nAChR-targeting radiotracers are scarce for imaging outside the thalamus, where the receptor is less densely expressed.
A team at Johns Hopkins University assessed the pharmacokinetic behavior of 18F-XTRA, a new PET imaging radiotracer for the α4β2-nAChR. The researchers tested the new radiotracer on a group of 17 adults and focused on extrathalamic regions of the brain. The research team found that 18F-XTRA rapidly entered the brain and distributed quickly.
“We present data using a new radiotracer with PET to characterize the distribution of the α4β2-nAChR in the human brain,” said Martin Pomper, MD, PhD. “The observed high uptake into the brain, fast pharmacokinetics, and ability to estimate binding in extrathalamic regions within a 90-minute scan supports further use of 18F-XTRA in clinical research populations. We also report the finding of lower 18F-XTRA binding in the hippocampus with healthy aging, which marks a potentially important finding from biological and methodological perspectives.”
The team said their findings will be important for future studies, especially in cases relating to neurodegeneration and aging, to monitor and assess changes in the human brain.
“Together, our results suggest that 18F-XTRA PET may be sufficiently sensitive to measure the hypothesized loss of α4β2-nAChR availability over aging, particularly in the hippocampus,” Dr. Pomper said. “This is a promising tool for the future study of changed cholinergic signaling in the brain over healthy aging that may be linked to changes in memory over the lifespan.”