Researchers have taken a major step towards developing a stem cell replacement therapy for debilitating muscle diseases.
A team from UCLA have developed a new strategy to efficiently isolate, mature and transplant skeletal muscle cells created from human pluripotent stem cells, which can produce all cell types of the body.
The findings could pave the way for a stem cell replacement therapy for muscle diseases including Duchenne Muscular Dystrophy, which impacts approximately 1 in 5,000 adolescent males in the U.S., and is considered the most common fatal childhood genetic disease.
The researchers used the natural human development process as a guide to develop ways to mature muscle cells in the laboratory and create muscle fibers that restore dystrophin, the protein that is missing in the muscles of Duchenne patients.
Muscles degenerate and become progressively weaker without dystrophin. Symptoms generally begin in early childhood and patients gradually lose mobility and typically die from heart or respiratory failure around age 20.
Scientists have attempted different methods that direct human pluripotent stem cells to generate skeletal muscle cells that can function appropriately in living muscle and regenerate dystrophin-producing muscle fibers.
In the current study, the researchers found that current methods produce immature cells that are not appropriate for modeling Duchenne in the lab or creating a cell replacement therapy for the disease.
“We have found that just because a skeletal muscle cell produced in the lab expresses muscle markers, doesn’t mean it is fully functional,” senior author April Pyle, an associate professor of microbiology, immunology and molecular genetics and member of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA, said in a statement.
“For a stem cell therapy for Duchenne to move forward, we must have a better understanding of the cells we are generating from human pluripotent stem cells compared to the muscle stem cells found naturally in the human body and during the development process,” he added.
After analyzing human development, the researchers found a fetal skeletal muscle cell that is extraordinarily regenerative and contains two new cell surface markers—ERBB3 and NGFR—which enabled the researchers to precisely isolate muscle cells from human tissue and separate them from various cell types to create using human pluripotent stem cells.
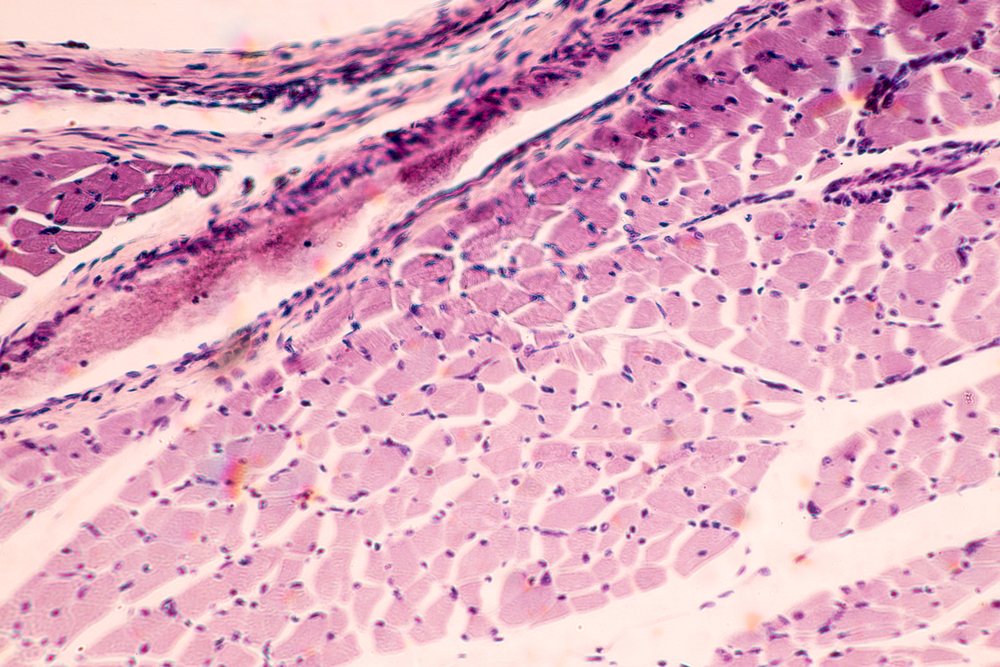
The researchers matured the cells to create uniformly muscle cells with fibers that were smaller than those found in real human muscle.
The team then discovered a specific cell signaling pathway called TGF Beta that needs to be turned off to enable generation of skeletal muscle fibers that contain the proteins that help muscles contract.
“Our long term goal is to develop a personalized cell replacement therapy using a patient’s own cells to treat boys with Duchenne,” Michael Hicks, lead author of the study, said in a statement. “So, for this study we followed the same steps, from start to finish, that we’d follow when creating these cells for a human patient.”
The study was published in Nature Cell Biology.