.jpeg)
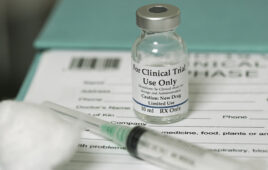
This photo shows vials containing AC5 in liquid and powder (undissolved) forms, which consists of self-assembling peptide technology used to stop bleeding. (Credit: Arch Therapeutics)
Advances in biotechnology have resulted in the explosive growth of biomaterials available to the modern surgeon. The need for better solutions is significant because bleeding occupies a large share of a surgeon’s mind and time—with constant repetition, when a cut is made, bleeding must be controlled.
Bleeding during surgical procedures also puts extra stress on patients’ organs, which can lead to other problems. Also, when patients bleed significantly during surgery, the visual field can become compromised for surgeons, and this can increase the risk of error. In addition, for patients on antithrombotics, there is an increased risk of bleeding. In a worst-case scenario, excessive bleeding can lead to the death of a patient.
Furthermore, bleeding during procedures can have an impact on the patient beyond the initial operation. It may lengthen his/her stay in the hospital as well as lead to abnormal healing, adhesions, hematomas and seromas after the procedure. And these challenges can lead to other problems, including infection and an even longer hospital stay, which can be clinically relevant to setbacks for the patient.
A need for new solutions
Interestingly, many of the tools doctors use to stop bleeding can cause their own challenges. Typically, to control bleeding during surgery, O.R. personnel initially rely on pressure, suture, staples and energy-based devices such as cautery. They then resort to a group of hemostatic agents such as gelatin, collagen, cellulose, polymers, thrombin and fibrin sealants. Yet many of these incorporate significant drawbacks.
In light of this, work is underway to develop products that have the potential to make surgery and interventional care faster and safer by using a novel approach to hemostasis and to control leaking. One approach is to develop products based on self-assembling peptide technology.
When applied as a liquid or spray to a wound, such a hemostatic device would locally self-assemble into a nanofiber structure that is a physical barrier on the tissue, mechanically sealing the wound in order to stop substances, such as blood, from leaking.
Desirable features and encouraging results to date
The self-assembling technology peptide platform is very promising. Based on testing to date, such a hemostatic device can have a number of characteristics that may make it user-friendly. For instance, it can be designed to conform to irregular wound geometry, not require cold or frozen storage, and be easily applied. Additionally, it would not need to be sticky or glue-like, which could enhance its utility in the setting of minimally invasive surgery. It can be transparent, and therefore help surgeons and other healthcare providers to maintain a clearer field of vision during a procedure. It could potentially be applied prophylactically so that additional bleeding is prevented just as it starts. Such a product would potentially represent a new generation of solutions for hemostasis and more.
Animal tests of a self-assembling peptide based hemostatic device under development have demonstrated rapid average time to hemostasis when applied to bleeding wounds in a variety of animal tissues. The technology can result in time to hemostasis of under 15 to 30 seconds. This is compared to much longer times, often from 80 seconds to significantly more than 300 seconds, when various control substances were applied.
The potential benefits could include better efficacy and outcomes, fewer risks, lower morbidity, less need for other resources, and faster throughput in the operating room. Perhaps most importantly, animal studies also support that such devices can work in the presence of antithrombotics, commonly known as blood thinners.
A recent clinical study to assess the safety and performance of one such hemostatic device in humans enrolled 46 patients, including 10 who were taking antiplatelet therapy. Each patient had bleeding wounds created as a result of the excision of two skin lesions under local anesthetic in the same setting. On a randomized basis, one lesion received treatment with the hemostatic device and the other received a control treatment consisting of standard therapy plus a sham. Each subject was followed-up for safety assessment both on Day 7 and again on Day 30, which marked the end of the patient’s participation in the clinical study.
The objectives of the study were to evaluate the safety and performance of the hemostatic device in patients scheduled to undergo excision of skin lesions on their trunk or upper limbs. The primary endpoint was safety throughout the surgical procedure and until the end of a 30-day follow-up period post procedure. Safety of the device was determined by monitoring for treatment-related adverse events. The primary objective was met, as the safety outcomes of both the device treatment group and the control group were similar. No serious adverse events were reported.
A secondary endpoint was performance as assessed by time to hemostasis. The median time to hemostasis of wounds in the treatment group was 41 percent faster than for those in the control group. These top-line data support that the hemostatic device was safe and performed as expected in the patients enrolled in the study throughout the completion of the patient assessments post-treatment.
Further analysis revealed that a subgroup of 10 patients who were taking a prescribed antiplatelet medication showed similar benefit from the hemostatic device as those not on antiplatelet medication. Specifically, the reduced median time to hemostasis of the treated wounds versus the control wounds was statistically significant for both the overall group of 46 patients and for the subgroup of 10 patients on antiplatelet therapy. Further, the median hemostasis for wounds treated with the device was less than 30 seconds for both the overall study group and for the subset of patients taking antiplatelet therapy.
Further such studies will improve our picture of the ability of a new generation of hemostatic devices to serve as an effective option for surgeons and their patients.
Terrence W. Norchi, M.D. is president & CEO of Framingham, Mass.-based Arch Therapeutics Inc. Arch’s flagship development stage product candidate, the AC5 Surgical Hemostatic Device, is being designed to achieve hemostasis in minimally invasive and open surgical procedures.