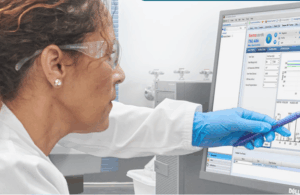
 Thermo Fisher Scientific released Chromeleon 7.4, a unified chromatography and mass spectrometry data system that supports single-quad, triple-quad, and HRAM instruments and adds centralized storage, remote access, audit trails, and e-signatures for GxP/21 CFR Part 11 compliance in biopharma, food safety, and environmental labs, according to a press release.
Thermo Fisher Scientific released Chromeleon 7.4, a unified chromatography and mass spectrometry data system that supports single-quad, triple-quad, and HRAM instruments and adds centralized storage, remote access, audit trails, and e-signatures for GxP/21 CFR Part 11 compliance in biopharma, food safety, and environmental labs, according to a press release.



