Scientists from the Tissue Engineering Group at the University of Granada (UGR) and the ibs.GRANADA Biomedical Research Institute have developed an artificial cornea based on fish scales. They published their research in Materials & Design.

Credit: University of Granada
Diseases that affect the cornea, the transparent part of the eye, are very difficult to treat because the cornea lacks blood vessels and has little capacity for regeneration and repair. Many patients can only be treated with a transplant. Only 1 in 70 patients worldwide who need corneal transplants receive donor tissue.
Now, researchers have created a biocompatible, resistant and transparent corneal implant from fish scales, potentially decreasing the dependence on organ donation, which is subject to waiting lists and shortages.
Multi-species comparison reveals carp as optimal candidate
Analysis of fish scales has demonstrated their potential effectiveness for corneal repair and regeneration, showing functional results in the laboratory and in animal tests. While previous studies examined fish scales for corneal applications, primarily focusing on tilapia with partial characterization, this is the first comprehensive multi-species comparison with full ex vivo and in vivo characterization.
The scales were obtained from red scorpionfish, salmon, white sea bream and European carp. The implants made from carp scales showed the least systemic reaction and the best optical properties. Implantations in rabbits demonstrated that the carp scales were biocompatible with the cornea. The corneas grafted with carp scale implants showed normal expression of corneal epithelium and stromal markers such as cytokeratin 3, crystallin alpha-A, p63, collagen, proteoglycans and lumican. The rabbits did not have any signs of rejection, inflammation or neovascularization.
Fish scales address limitations of existing bioengineered alternatives
The human cornea is composed of three layers: the outer stratified epithelium, the stroma and the inner corneal endothelium. The preservation of this structure is essential for light to reach the retina. Many conditions affecting the cornea cause a severe loss of vision or blindness.
Other bioengineered corneal tissues have been developed, including ones made of fibrin, alginate, collagen, methacrylate and polyethylene glycol-polycaprolactone. However, all of these showed limitations such as deformability, mechanical weakness and low integration with native tissues.
Fish scales, besides being easily obtained from the fishery and aquaculture industry, are dermal-origin structures that play a protective role for the animals. The scales are usually transparent and mechanically resistant. Most scales consist of a mixture of organic compounds, including type-I collagen. The structure of these scales is able to partially reproduce the structure of collagen within the human cornea.
Unlike synthetic prostheses, fish scales enable the patient’s own epithelial cells to grow and integrate. The collagen-rich strcuture naturally mimics human corneal architecture without requiring complex synthesis.
The new study analyzed four types of scales from four different species ex vivo and in rats and rabbits to determine their biocompatibility, functionality and potential usefulness.
The fish scale implants were evaluated by histological analysis and HE staining, which revealed the differing thicknesses of the scales as well as the number of layers of material within them. DAPI staining was used to identify the cell populations of the scales, showing that salmon and sea bream had fewer cells than red scorpionfish and European carp.
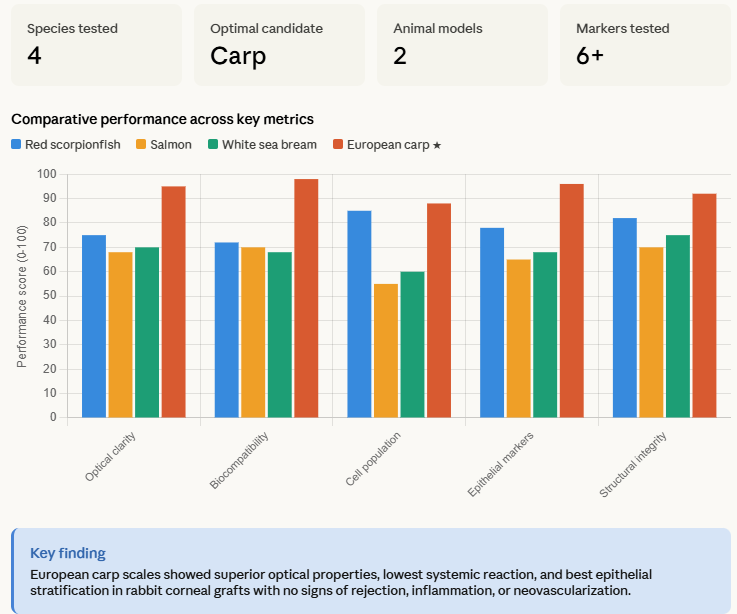
Data from Materials & Design
Industrial byproducts provide low-cost, scalable raw material
Fish scales are industrial byproducts, accounting for about 4% of global fish processing waste, which totals 7.2 to 12 million tons annually, making them easily obtained for corneal implants. The low-cost of the materials could make this treatment accessible in developing coutnries were organ donor shortages are most severe.
The research team emphasizes that the implant manufacturing process is “reproducible and scalable.” The next development stages include cleanroom production capabilities to meet regulatory standards for medical devices.