Image courtesy of William Grover/UCR
At least 1 in 10 medicines in low- and middle-income countries are substandard or falsified. A 2023 UNODC assessment estimated 267,000 deaths per year from falsified antimalarials alone in sub-Saharan Africa, with nearly 170,000 more from counterfeit antibiotics.
In the U.S., the problem is smaller in scale but growing: the CDC has warned about counterfeit medications sold through disreputable online pharmacies, and gray markets for popular GLP-1 receptor agonists and Botox have produced watered-down or illicit versions that have caused serious injuries and deaths.
Researchers at the University of California, Riverside report a low-cost counterfeit-drug screening tool built around an infrared sensor used in line-following toy robots. In proof-of-concept testing, the method correctly identified about 90% of 96 pills from 32 drug products; the current hardware is described as roughly $33, with the university saying future manufacturing cost could fall below $30 and potentially to $5. The full design is open source, detailed in a paper published in Analytical Chemistry.
How dissolution fingerprinting works
The idea is straightforward: legitimate pills from the same product dissolve in water in a consistent, reproducible way because manufacturers tightly control formulation, compression, and coating. Counterfeit pills, made with different ingredients or processes, dissolve differently.
William Grover, an associate professor of bioengineering at UC Riverside, and his team built a low-cost optical dissolution monitor around a reflectance-based infrared sensor like the ones used in line-following robots. As a pill dissolves, the sensor tracks changes in the sample’s optical properties and converts them into a digital “disintegration fingerprint” that can be compared with a known authentic reference.
“If it’s a legitimate medicine, the manufacturer made every pill identical enough that they’ll all behave roughly the same way when they dissolve,” Grover said.
So if you test a suspect pill, and it dissolves at a different rate than the real thing, this suggests the suspect pill is counterfeit.
The team tested the system on more than 30 medications, including antibiotics, vitamins, prescription opioids, and over-the-counter painkillers. Roughly 90% could be correctly identified from their dissolution fingerprints. The method was sensitive enough to distinguish Bayer aspirin from store-brand aspirin, and even U.S. and Canadian versions of the same product.
 What already exists for counterfeit drug detection
What already exists for counterfeit drug detection
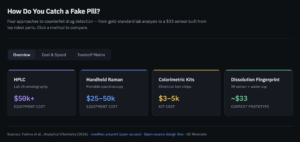
The current gold standard for pharmaceutical authentication is high-performance liquid chromatography, or HPLC, which can identify and quantify active ingredients with high precision. But it requires a laboratory, consumables, and trained personnel. It is not a field tool.
Handheld spectroscopic devices have made field screening possible. Systems such as Rigaku’s Progeny handheld Raman spectrometer and Thermo Fisher Scientific’s TruScan enable rapid, non-destructive identification of pharmaceutical compounds outside the lab. But they still require specialized hardware, which can place them beyond the reach of community health workers, small pharmacies, and NGOs operating in regions most affected by counterfeit drugs.
Cheaper colorimetric test kits, such as the Minilab system promoted by the Global Pharma Health Fund, can help detect missing active ingredients. But they generally cannot measure concentration precisely or reliably distinguish more sophisticated counterfeits that contain some active ingredient at reduced levels.
The UCR dissolution fingerprinting approach falls between these categories. It cannot identify specific chemical compounds like spectroscopy or HPLC can. What it can do is flag pills that behave differently from a known legitimate reference. At sub-$30 cost, that tradeoff may be exactly right for the highest-need settings.
Where this could go
“Watered-down or illicit versions of drugs like Botox or popular GLP-1 receptor agonists have caused serious injuries or death,” Grover noted, pointing to the domestic relevance of counterfeit drug detection. The CDC has warned that people ordering what they believe to be prescription medications from disreputable online pharmacies frequently receive fakes. But the team’s stated future goal is applying the technology to fake antimalarial drugs in tropical regions, where the problem is most acute and the resources for detection are thinnest. A 2023 UNODC threat assessment estimated 267,000 deaths per year from falsified antimalarials alone in sub-Saharan Africa, with nearly 170,000 more from counterfeit antibiotics used to treat childhood pneumonia
The 90% accuracy figure, while promising for a proof of concept, will need to improve for real-world deployment. A 10% false-negative rate in counterfeit detection is a significant gap. The open-source design creates a path for other research groups to iterate on the hardware and the fingerprint-matching algorithms, but moving from lab validation to a ruggedized field tool is a different engineering challenge entirely.
Still, this is a novel approach to a problem that existing technology has failed to solve at the price point that matters most. Others have used dissolution rates to assess drug legitimacy before, but Grover’s team is the first to convert that process into a digital signature — a “disintegration fingerprint” — using a sensor that costs as little as $5. The clever part isn’t the sensor; it’s the realization that dissolution kinetics encode enough information about a pill’s formulation to tell you whether it’s real. That insight, paired with a component cheap enough for a toy robot, is worth more than another $30,000 handheld spectrometer that never makes it to the clinic that needs it.