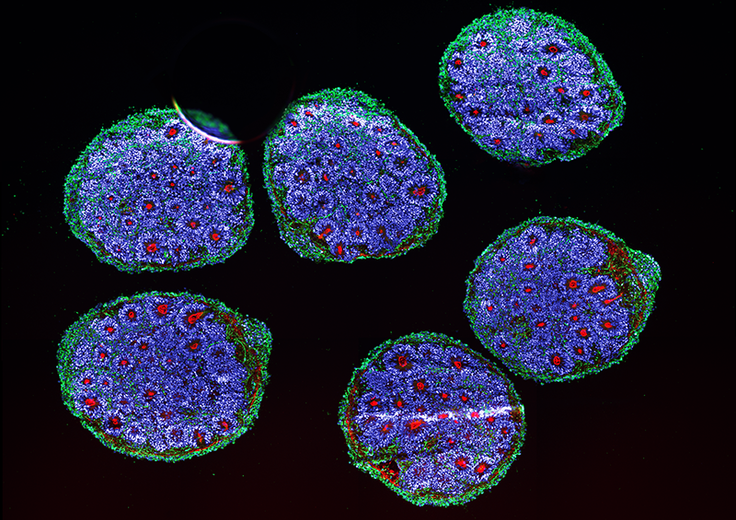
Human brain organoids grown from stem cells, showing neurons and dendrites (green), forebrain cells (blue), and tight junctions between cells (red). In a 2022 study, researchers at the Broad Institute tracked organoid development over six months and found the same cell types arose in the same order and expressed the same genes as cells in the developing human embryo. Credit: Noelia Anton Bolanos and Irene Faravelli/Broad Institute
For decades, the loudest voices against animal testing in drug development belonged to animal rights groups and organizations like PETA. The pharmaceutical industry largely tuned them out while committing to the three Rs, which include Replacement, Reduction and Refinement of preclinical testing processes. So it was striking to see FDA encouraging drug developers to plan on moving beyond animal testing. “God did not make animals on planet Earth for us to abuse and torture,” FDA Commissioner Marty Makary told attendees at a joint FDA-NIH workshop in July 2025.
The push has drawn mixed reactions while also spurring interest in alternative testing methods, spanning organoids and organs-on-a-chip, among others. In March 2026, FDA issued draft guidance around new approach methodologies, while Europe has spent years building validation infrastructure for non-animal methods and Japan’s PMDA is putting organoids and microphysiological systems into its regulatory-science agenda. NIH is putting serious money behind human-based models.

A lung-on-a-chip device developed at the Wyss Institute at Harvard. The transparent chip, roughly the size of a memory stick, uses microfluidic channels to recreate the mechanical and biological environment of a human lung, including breathing motions and air-blood tissue interfaces. In 2022, Cantex Pharmaceuticals included data from a Wyss lung chip in an IND filing for a repurposed COVID-19 drug, among the earliest cases in which organ-chip data were included in a regulatory submission to FDA. Credit: Wyss Institute, Harvard University
Animal testing itself has something of a mixed track record at predicting safety and efficacy. For instance, in 1993, a hepatitis B drug called fialuridine sailed through toxicology studies in mice, rats, dogs and monkeys with no sign of liver problems. In humans, seven of 15 trial participants developed acute liver failure. Five died. Two survived only because they received emergency transplants. A National Academy of Sciences review later concluded that the animal data provided no indication the drug would be hepatotoxic in people.
Similarly, in 2006, the antibody TGN1412 looked safe in rodents and primates at high doses. Six volunteers in London received an infusion at one five-hundredth the dose found safe in animals. Within hours, all six were in intensive care with multiorgan failure.
Animal testing may predict what a compound does in a human body. It may not. And when it doesn’t, the consequences land on patients.
The caveat is that a weak animal model does not automatically make a human-derived model decisive. The strongest evidence is still context-specific. A 2022 study of 870 Liver-Chips tested against 27 benchmark drugs reported 87% sensitivity and 100% specificity for drug-induced liver injury, and patient-derived tumor organoids have predicted treatment response in some cancer settings. But the field is still wrestling with immune complexity, long-term culture stability, reproducibility, vascularization and standardization. The question is where these models are predictive enough to change the decision.
Organoids are one attempt to close that gap. Grown from patient-derived tissue or stem cells rather than immortalized cell lines, they recapitulate more of the biological complexity that animal models and conventional cell cultures miss. “We started with cell lines, moved into more complex cell lines and different structures, looked at single cell suspension type of systems, expanded it to look at 2D cultures, and then 3D cultures as well, looking at tumors and then organoids,” said Dan Benjamin of Immuto in an interview with R&D World. “This is more like a patient-derived model that includes a lot more heterogeneity. It really captures a lot of the native biology that you would see from an actual patient.”
The demand for that kind of biological fidelity is growing fast, in part because the drug discovery pipeline is getting crowded from the other end. AI-driven drug design has accelerated the pace at which pharmaceutical companies can churn out candidate molecules, but the compounds still need to be tested against living tissue. “They have so many drugs from these AI drug discovery companies, they kind of oversaturated,” said Parmita Mishra, CEO of Precigenetics, a cellular imaging company based in Silicon Valley, in an April 2026 interview. “Their entire bottleneck has become testing.” The economics sharpen the point: a single primate toxicology study can run $1.5 million per compound and take three years to set up, Mishra said, while cell-based screening platforms can run the same assessment in the five- to six-figure range.
Organoids are one example of what the field broadly calls New Approach Methodologies, or NAMs. At the same July 2025 workshop, Nicole Kleinstreuer, acting NIH deputy director for program coordination, framed the agency’s commitment in structural terms. “We are really shifting NIH’s investment to prioritize technologies that are rooted in human biology,” she said, citing organoids, microphysiological systems, 3D bioprinting and computational modeling. NIH had already announced plans to establish the Office of Research Innovation, Validation, and Application (ORIVA), to coordinate the development and regulatory translation of those methods.
Among NAMs, organs-on-a-chip, also known as microphysiological systems, have the longest regulatory paper trail. In 2021, Sanofi filed what is believed to be the first IND application using organ-chip data as the sole efficacy evidence, relying on a human-on-a-chip model built by Hesperos to support its CIDP therapy sutimlimab. No animal efficacy studies were conducted. FDA authorized the trial. Since then, Cantex Pharmaceuticals included lung-chip data from the Wyss Institute in an IND for a repurposed COVID-19 drug, and in September 2024, CDER accepted the first organ-on-a-chip technology into its ISTAND qualification pilot: a Liver-Chip designed to predict drug-induced liver injury.
Those precedents are real, but regulatory traction still depends on matching the right tool to the right problem. “The biggest concept here is that if you want something to be used, you have to understand the context of use,” said Warren Casey of NIH/NIEHS at the July 2025 workshop. FDA acceptance is center-specific, Casey noted: “CDER has different requirements than CBER, just different than CDRH. So if you want those centers to actually use something, you have to start with them, understand what they really need, and then convey that to the test method developers.”
That context-of-use specificity is also what makes validation slow. Casey said the process can still take “six to 10 years,” and warned that the field has already spent “millions and millions of dollars into tests that nobody was using” because developers built methods before confirming what regulators would accept. Organs-on-a-chip have moved furthest because they now have named IND precedents, a defined ISTAND pathway and concrete contexts of use. The broader leap from mice to human biology will likely depend on whether organoids, chips, computational models and imaging platforms can move through that same use-specific validation funnel.
Still, the institutional momentum is accelerating faster than Casey’s timelines might suggest. In a JAMA Perspective published April 20, 2026, acting CDER director Tracy Beth Høeg and colleagues, including Makary, outlined the FDA Roadmap’s first-year accomplishments: draft guidance to reduce or eliminate routine six-month primate toxicology studies for certain monoclonal antibodies (potentially sparing hundreds to more than a thousand nonhuman primates a year), a permanent ISTAND qualification pathway, a searchable public database of accepted NAMs, and a formal FDA-NIH memorandum of understanding. The piece framed the shift as “a paradigm shift away from asking for animal data to determining what data are necessary for human safety and how these data can best be obtained.” The question for the field is whether the validation infrastructure can keep pace with that ambition.
“Fifty years from now, we will not be testing in animals, and I don’t think we’ll be testing in humans either. I think all of drug discovery will be done virtually,” said Robert DiFazio of Parallel Bio in a January 2026 interview. “Might be running it in something like organoids as the final step, maybe doing a small human trial. But the whole thing should only take, like, a year.”




Tell Us What You Think!
You must be logged in to post a comment.