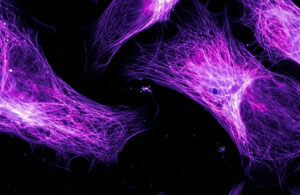
High-resolution fluorescent imaging of cellular structures. (Source: Precigenetics)
Suppose the only way to search for life other planets was to vaporize them first, then sift through the debris. “Imagine if you had to literally nuke planets to figure out whether there’s alien life there,” said Parmita Mishra, founder and CEO of Precigenetics, a San Carlos, California-based real-time cellular imaging startup. “We would never know anything. That’s what we do to cells every single day.”
A core limitation in cell profiling is that many high-dimensional methods are destructive. As a Raman2RNA paper in Nature Biotechnology noted, single-cell RNA sequencing and related profiling assays are “inherently destructive.” This dynamic makes it a challenge to track the temporal dynamics of living cells over time. Mishra argues that biology has evolved at a slower rate than several other scientific fields. “We have rovers on Mars. We have the James Webb Space Telescope, yet in biology we are still bottlenecked by measurement tools that kill the sample,” she said. Precigenetics says its platform is built for that problem, using a proprietary optical measurement system to generate what it calls “real-time chemical movies of living cells.”
Founded in 2024, Precigenetics currently has six full-time employees plus interns, incubated at MBC BioLabs in San Carlos. The company has backing from O’Shaughnessy Ventures, Chaotic Capital and 1517 Fund, and is pursuing a seed round that Mishra says she hopes will reach $7 million to $10 million.
Toward a ‘telescope’ for understanding living systems
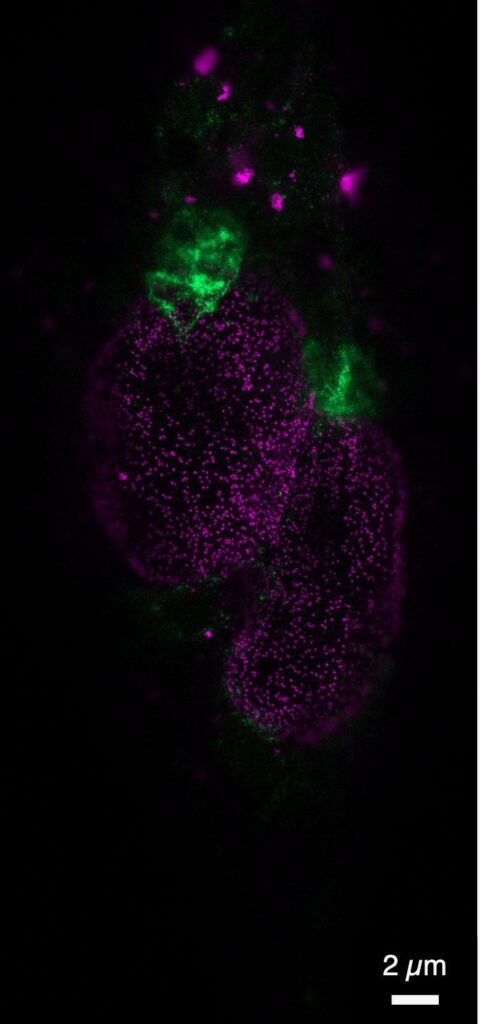
Subcellular resolution image from Precigenetics.
The foundation for Precigenetics would come while Mishra was studying at the Epigenetics Institute at Penn Medicine. There, she contributed to single-cell hydroxymethylation research, but left the PhD track out of frustration with the reliance on animal models misaligned to human disease, and a lab workflow bottlenecked by a genome sequencer. “We were studying Alzheimer’s disease in mice, and that made no sense to me,” Mishra said. “A mouse lives for two years. A mouse does not really get Alzheimer’s disease.”
Another turning point came with MIT’s Raman2RNA project, which demonstrated that label-free noninvasive Raman images of live cells could be computationally mapped to RNA expression profiles. Mishra said the work pushed her toward a different question: why not keep cells alive under tightly controlled conditions, including physiological temperature and 5% CO₂ via microfluidics and image them continuously using optics borrowed from semiconductor manufacturing?
In other words, Precigenetics is trying to use the same nanometer-scale optical precision found in semiconductor imaging on living cells. Mishra said the trick is to keep the cells in what the company calls an “artificial lung,” with controlled temperature, roughly 37°C for most cells and about 36°C for skin cells, plus 5% CO2 delivered through microfluidics. Under those conditions, she said, the company can apply semiconductor-style optical imaging to biological cells.
In Precigenetics’ system, the setup involves a room-scale system with femtosecond lasers and custom microfluidic chips. Rather than recording red, green and blue for each pixel, the system collects a full spectrum, which is why the data volume per cell runs into gigabytes. The data can run roughly two to three gigabytes every two hours.
Mishra said the spectral approach reveals differences that conventional microscopy cannot. Two cells can appear identical under a standard microscope while one is dying and the other is adapting, a distinction visible only in their chemistry. The system captures those chemical shifts continuously, producing what amounts to video-rate data from living human cells.
Mishra argues that better live-cell measurement will shape how useful AI becomes in drug discovery. Here, Mishra invokes Derek Lowe, director in chemical biology and therapeutics at Novartis Institutes for BioMedical Research and author of the long-running In the Pipeline blog, who has argued that the industry’s hardest problems, picking the right targets and anticipating human toxicity, are “almost inversely proportional to the ability of AI to solve them.” In other words, generative chemistry can propose compounds all day, but if you cannot observe what those compounds actually do to living human cells over time, you are still guessing.
For Mishra, that hardware-heavy architecture is the point. She argues that drug discovery needs the same kind of engineering step-change that once transformed sequencing. “The one time we saw costs truly collapse and efficiency truly rise was in sequencing, and that happened because of optics, computation, engineering and acquired hardware IP,” Mishra said. Sequencing is “great for diagnostics, but for drug discovery we need something else,” she continued. “We need the equivalent of a telescope for understanding living systems.”
Turning living biology into computable data
Mishra notes that many biotech companies are still focused on single targets at a given time. “They label a target, make it glow, and ask whether a drug binds to it,” she said. “Our view is that if you remove fluorophores and labels, you are no longer locked into one target. You can begin to see toxicity, resistance and all the things happening outside that single target in the entire cell.”
In turn, she says that the ability to keep the cell alive will yield substantially more complicated models. “You can make liver models. You can use patient cells,” she said. “One of the projects that is especially emotional for us involves a rare-disease organization.” Mishra continues: “There was one child with a disease, and those were the only cells they had. Now there’s another child who needs a drug. They had no practical way to screen candidates until they realized they could send those cells to us and we could be far more economical with them because we don’t keep killing the sample.
Mishra also argued that the regulatory climate is shifting in favor of approaches like this. The FDA Modernization Act 2.0, signed in late 2022, removed the longstanding statutory mandate that all new drugs be tested on animals before human trials. More recently, the agency has moved faster: Commissioner Makary’s April 2025 roadmap began laying out a phaseout of animal testing for some drug classes, and in March the FDA released broader draft guidance on validating new approach methodologies in drug development. The NIH has moved in the same direction, saying in July 2025 that it would no longer issue funding opportunities focused exclusively on animal models of human disease.
Against that backdrop, Mishra said, “With the FDA talking more seriously about new approach methodologies and reduced reliance on animal models, some companies feel they can finally pursue disease areas and target classes they were not set up to pursue before.” She added that “there’s also growing interest in areas where animal models have been especially limiting, including women’s health and autoimmune disease,” and argued that “the recent changes around modernization and new approach methodologies matter.”
Biology still has its SpaceX moment ahead of it. —Mishra
Mishra said the company’s near-term commercial plan is built around that opening. Precigenetics is starting with toxicity screens, initially focused on liver metabolism, and targeting compound classes like antibody-drug conjugates and mRNA-related programs where conventional binary assays often miss the full picture. The company is building what it calls Cleopatra, a toxicity foundation model that Mishra described as an attempt at “an AlphaFold-like moment, but for actual toxicity mechanisms.”
Meanwhile, the pharmaceutical patent cliff is forcing companies to search for first-in-class targets rather than me-too compounds, the GLP-1 and peptide wave has reshaped investment priorities, and AI researchers are increasingly drawn to biology. “Biology still has its SpaceX moment ahead of it,” she said.
About nine out of ten drug candidates continue to fail in clinical trials, a success rate that has barely improved in decades, with toxicity alone accounting for roughly 30% of those failures. The entire global pharmaceutical industry is projected to spend around $308 billion on R&D in 2026. The same year, just five technology companies plan to spend between $660 billion and $690 billion on AI infrastructure. Much of that is directed at advertising, recommendation engines and social media optimization.
Mishra wished more of the funding would flow into healthcare. “The thing that keeps me up at night,” she added: “I know the power of AI, and I do not want to reach a date 10 years from now where we have recommendation algorithms that keep us addicted to Netflix, and little of that compute is actually going into curing cancer.”



Tell Us What You Think!
You must be logged in to post a comment.